Jesse Rowley and Dr. Gregory Burton, Microbiology
Introduction
Human Immunodeficiency Virus (HIV), the causative agent of AIDS, has eluded successful treatment since its discovery in the early 1980s. The prospect of successful treatment came with the advent of Highly Active Anti-Retroviral Therapy (HAART) which has reduced plasma virus replication to undetectable levels in many patients. Although virus may be maintained below detectable levels for years by treatment, complete viral clearance has not been achieved. “Hidden” reservoirs of non-replicating or slowly replicating virus escape rigorous drug treatment. When the patient is taken off of anti-retroviral therapy, plasma virus levels will return to pre-therapy levels and HIV will continue the course of infection.
Previous research has indicated that Follicular Dendritic Cells (FDCs) of secondary lymphoid tissues may act as a reservoir for HIV. FDCs trap and protect antigen/antibody complexes and display them on their surface in maintenance of the memory response. During the course of HIV infection, FDCs become heavily laden with virus (1). Recent research done in our lab has shown that in a murine model, this trapped virus may remain infectious for long periods of time (2). Further, although plasma virus levels decline drastically during HAART treatment, image analysis reveals that a stable pool of virions remains associated with FDCs (3). In vitro, HIV bound to human FDCs remains infectious for up to 25 days as measured by HIV transfer from FDCs to T cells (2).
HIV is a rapidly evolving virus that mutates frequently under the selective pressures of the immune system and/or drug treatment. Because FDCs trap virus during infection, the HIV quasi-species found on the surface of the FDC contains evolutionarily distinct forms of virus dependant on the time of trapping. In this study we determine the genetic relationships among HIV species trapped on FDC and within lymph node T-cells. HIV POL, which contains PRO and RT genes, is a principle target of many antiviral drug regimens. In the present study we compare the genotypic relationship of HIV POL found on the surface of lymph node FDCs isolated from an HIV infected patient (621) and HIV POL found in patient 621 spleen CD4 T cells.
Methods
Spleen and Lymphoid tissues from HIV infected patient 621 who had been treated with HAART were provided by Johns Hopkins University. Lymph node T cells and Spleen FDCs were isolated using previously described procedures (1). cDNA synthesized from HIV RNA isolated from FDCs, and DNA isolated from CD4 T cells was amplified by nested set PCR. Outer and inner primers to RT 5’ and Pro 3’ were chosen to amplify the entire POL gene. PCR amplification products were cloned into plasmid vector NPCR3.1 using Eukaryotic TA cloning Kit (Invitrogen) and transformed via heat shock into STBL2 cells. Cells were cultured overnight in LB, and streaked onto LB plates. Colonies were screened for pol inserts by gel electrophoresis. Pure DNA was isolated using Qiagen filter kits and sequenced using Big-dye (Applied Biosystems). Sequences were manually edited in SEQUENCHER (Gene Codes Corp., Ann arbor, Mich). Nucleotide alignments were generated in CLUSTAL X (4) and then edited and gap-stripped in MacClade (5). The best model for evolution of this data set was determined in MODELTEST (6). A maximum likelihood tree was created in PAUP* (7).
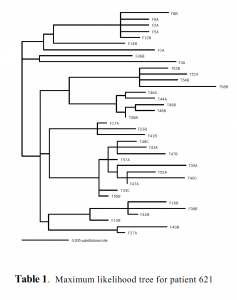
Results
18 clones of HIV POL from LN FDCs cDNA and 19 clones of HIV POL from Spleen CD4 T cell DNA were sequenced and combined into a maximum likelihood tree (Table 1). Because this work represents only a small portion of a more extensive study, at this point little can be determined from the data at hand. We are still attempting to clone POL for HIV cDNA and DNA of Spleen FDCs and LN CD4 T cells, and plan to sequence the ENV gene for patient 621. Other undergraduates in the lab have sequenced portions of HIV isolated from 2 other HIV infected patients. We will use this information to evaluate the contribution of FDCs as a reservoir for drugsensitive versus drug-resistant HIV species. Understanding the genetic nature of the FDC reservoir will be important to eventual viral eradication.
References
- Heath, S.L., Tew, J.G., Szakal, A.k., and G.F. Burton. 1995. Follicular Dendritic cells and human immunodeficiency virus infectivity. Nature 377:740.
- Smith, B.A., Gartner, T., Liu, Y., Perelson, A.S., Stilianakis, N.I., Keele, B.F., Kerkering, T.M., Ferreira-Gonzalez, A., Szakal, A.K., Tew, J.G., and G.F. Burton. 2001. Persistence of Infectious HIV on Follicular Dendritic Cells. J. Immunology. 166: 690-696.
- Haase, Ashley T., Henry, Keith, Zupancic, Mary, Sedgewick, Gerald, Faust, Russell A., Melroe, Holly, Cavert, Winston, Gebhard, Kristin, Staskus, Katherine, Zhang, Zhi-Qiang, Dailey, Peter J., Balfour Jr., Henry H., Erice, Alejo, Perelson, and S. Alan. 1996. Quantitative Image Analysis of HIV-1 Infection in Lymphoid Tissue. Science 274: 985-989.
- Thompson, J.D., D.G. Higgins, and T.J. Gibson. 1994. CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, positionspecific gap penalties and weight matrix choice.
- Maddison, D.R., and W.R. Maddison. 1992. MacClade—analysis of phylogeny and character evolution—version3. Sinauer Associates, Inc., Sunderland, Mass.
- Posada, D., and K.A. Crandall. 1998. MODELTEST: testing the model of DNA substitution. Bioinformatic 14:817-818.
- Swofford, D.L. 1999. PAUP*: phylogenetic analysis using parsimony, edition 4.0. Sinauer Associates, Sunderland, Mass.