Chris Meinhart and Dr. Laura C Bridgewater, Microbiology
Damage to articular cartilage found in synovial joints can occur both by trauma and disease processes. Cartilage, because of its avascular nature and low cellularity, has very poor regeneration capabilities. It is therefore important to explore new and effective cellular therapies to facilitate the repair of diseased or otherwise damaged cartilage.
It has been shown that bone marrow derived adult mesenchymal stem cells (MSC’s) have the ability to differentiate down several cell lineages including a chondrogenic one. This chondrogenic differentiation, which is the developmental differentiation of cells to become cartilage cells, or chondrocytes, was first induced using a protocol with a defined chondrogenic media by Johnstone et al.¹ Later modifications to this protocol by other investigators have had the effect of increasing the expression of the chondrocytic phenotype. These expressed characteristics are found in the extracellular matrix secreted by the cells, including increased amounts of proteoglycans, collagen type II and XI.
Hepatic oval cells, originally thought to be the progeny of hepatic stem cells, have recently been shown to be derived from a bone marrow precursor cell.² Other circumstantial evidence exists supporting this idea. These cells also contain markers showing their close relationship to hematopoietic cells.³ This information suggests that hepatic oval cells possess a more pluripotent potential than originally thought. This newly understood potential makes them an excellent candidate for further chondrogenic studies. An improved protocol for the differentiation of MSC’s has been defined by Sekiya et al.4
Following the methods defined by Sekiya et al.4 approximately 200,000 oval cells were centrifuged in 15-ml polypropylene conical tubes. The pellet formed was cultured in chondrogenic media consisting of high-glucose DMEM supplemented with 500 ng/ml BMP-6 as well as 10 ng/ml TGF-ß3, 10-7 M dexamethasone, 50 μg/ml ascorbate 2-phosphate, 40 μg/ml proline, 100 μg/ml pyruvate, and 50 mg/ml ITS+ Premix. Media was changed every 2-3 days.
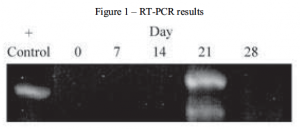
While many different types of collagen have been identified and are used throughout the body in various tissues, collagen type II is specific to a chondrocytic phenotype. To determine the extent of collagen II expression, chondrogenic pellets were digested and the total RNA isolated. After the specific isolation of mRNA and conversion to cDNA, PCR amplification was performed. The amplification products were then resolved by electrophoresis. This process was performed at 0, 7, 14, 21 and 28 days during pellet culture. RNA from rat chondrosarcoma (RCS) cells were used as a positive control for the expression of collagen type II. As demonstrated in Figure 1, chondrogenic differentiation did not occur during this experiment.
As an additional indication of chondrogenic differentiation we attempted histological examination of paraffin sections. Our intent was to identify proteoglycans, a marker of early stage chondrogenesis, by staining pellet sections with Safranin O. Unfortunately, the pellets were not stable enough to survive the paraffin embedding process. This is most likely due the lack of collagen and proteoglycan synthesis by the oval cells.
As these previous attempts at chondrogenesis were unsuccessful, we have taken a new approach. The cell signaling pathways of the cytokines TGF-â5 and BMP6 have recently been described. As these two cytokines are critical components of chondrogenic differentiation studies, they are of particular importance in understanding what is and isn’t occurring during chondrogenic differentiation. Both pathways are quite similar and describe the interaction and phosphorylation of various proteins called SMAD’s. It is our intent to retry our previous experiments and include RT-PCR analysis in an attempt to identify if and when these SMAD proteins are present during this process. As a control for these experiments human mesenchymal stem cells will also be grown in culture and analyzed as previously described. Our aim is to identify which components of these pathways were not present in oval cells and presumably identify a possible solution to inducing differentiation.
In the pursuit to discover the mechanisms of chondrogenic differentiation, the answers to important questions lie waiting. New and effective therapies to repair damaged cartilage could result from this work. The ability to successfully administer these treatments is, however, dependent on the level of understanding of chondrogenic processes and to a large extent on reliable sources for chondrogenic differentiation. While it has been shown that MSC’s have a chondrogenic potential, the identification of additional chondrogenic sources will have significant importance. For these reasons, it remains a critical goal to describe chondrogenic processes in other cell lines.
References
- Johnstone, B., Hering, T.M., Caplan, A.I., Goldberg, V.M., and Yoo, J.U. (1988) Exp. Cell Res. 238, 265-272
- Petersen, B.E., Bowen, W.C., Patrene, K.D., Mars, W.M., Sullivan, A.K., Murase, N., Boggs, S.S., Greenberger, J.S., Goff, J.P. (1999) Science 14;284(5417):1168-70
- Petersen, B.E., Goff, J.P., Greenberger, J.S., Michalopoulos, G.K. (1998) Hepatology 27(2):433-45.
- Sekiya, I., Colter, D.C., Prockop, D.J. (2001) Biochem Biophys Res Commu. 8;284(2):411-8.
- Fujii, M., et al., (1999) Roles of bone morphogenetic protein type I receptors and Smad proteins in osteoblast and chondroblast differentiation. Mol. Biol. Cell., 10, 3801-3813.
- Kawabata, M., and Miyazono, K., (1999) Signal transduction of the TGF-â superfamily by Smad proteins. J. Biochem. (Tokyo), 125, 9-16.