Jeremy F McBride, Rachelle R Olsen and Dr. Kim L O’Neill, Microbiology and Molecular Biology
Angiogenesis, the development and differentiation of new blood vessels from the pre-existing vasculature, is a fundamental physiological process involved in growth, development, and wound healing. Loss of control over the biological molecules which regulate this process can lead to pathologic angiogenesis, or neoangiogenesis, a process associated with such diseases as retinopathies, psoriasis, arthritis, and most importantly, cancer.
Over the last thirty years, research has shown that angiogenesis is absolutely required for a cancerous tumor to grow beyond a few millimeters in size. The new vasculature provides the tumor with not only a source of nutrients and a waste outlet, but also with a transport system which can facilitate metastasis, the establishment of additional tumors at distal regions. Since angiogenesis is absolutely required for tumor growth, one major topic in current research is the discovery and characterization of anti-angiogenic drugs, several of which are in clinical trials.
We decided to study an extract of Convolvulus arvensis after an initial report of a woman suffering from ovarian cancer whose cancer went into complete remission following consumption of this plant. Since preliminary studies showed that the anti-cancer properties of C. arvensis may be due to anti-angiogenesis, we decided to use both in vitro and in vivo models to investigate the mechanisms by which it inhibits angiogenesis.
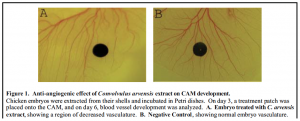
We began studying the anti-angiogenic properties of C. arvensis extract using the Chorioallantoic Membrane (CAM) Assay. This in vivo model using chick embryos is one of the most-used systems for studying angiogenesis, as it provides real tissue barriers, blood vessels, and basement membranes. The results we obtained, which suggest that C. arvenesis has antiangiogenic properties, are shown below in Figure 1. Figure 1.
While C. arvensis appeared to inhibit angiogenesis, it was difficult to obtain definitive results from this assay due to three factors. First, we lacked the ability to quantify the degree of blood vessel development directly surrounding the treatment patch. Second, we had difficulty keeping the patches precisely in place as the embryo developed. Third, there was a high amount of natural variation in the blood vessel arrangements within the CAM.
At this point, we modified our research to focus specifically on the effects of C. arvensis on endothelial cells (ECs), the cells which line all blood vessels and are the first to respond to an angiogenic stimulus. Once ECs were obtained, we planned to use the Alamar Blue assay, flow cytometry, RICAM assay, Comet Assay, and a modified CAM Assay to examine proliferation, cell cycle distribution, migration, apoptotic index, and angiogenic ability, respectively.
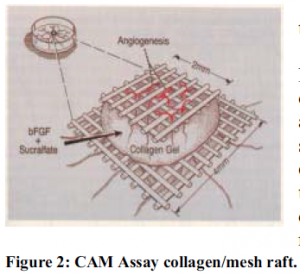
The EC line we chose to study, E.A.hy926, was generously provided by Dr. Cora-Jean Edgell, of the University of North Carolina. After freezing down several vials of cells, we began working on the Alamar Blue assay and a modified CAM assay (Figure 2). In this assay, a collagen raft containing a known angiogenesis inducer, such as bFGF or VEGF, is used to induce vertical blood vessel formation. Embryos are treated with collagen rafts containing either the inducer alone, or both the inducer and a suspected inhibitor. Results are quantified by the percentage of squares in the top piece of nylon mesh which contain newly formed blood vessels.
The primary problem we had was that we were unable to get the collagen solution to solidify. We suspect that the UV treatment that was used to sterilize the solution probably also destroyed its ability to polymerize.
Meanwhile, we set up a cell dilution series to determine optimal densities to use in the Alamar Blue assay. In this assay, cells are incubated in media containing Alamar Blue, a substance which can act as a terminal electron acceptor in the electron transport chain, and which changes color, from blue to red, as it is reduced. The amount of color change is determined using a plate reader, and can be correlated to the rate of cell proliferation.
While we expected the wells to begin changing color within approximately 4-6 hours, no substantial color change was observed until 48 hours later. From these results, we knew the cells were either plated at densities much too low for this assay, or that our cells were proliferating very slowly. Within a week, most of our cells were no longer proliferating, a response that we did not suspect from an established cell line. We thawed out some additional cells, but each time they were contaminated a day later, despite treatment with antibiotics. It is possible that the cells were contaminated when they arrived, since these initial cultures were frozen down so early. Since we no longer had a source of ECs, we were unable to test our hypothesis using the approach outlined above.
Since this time, we have obtained some HUVECs (human umbilical vein endothelial cells), and we hope to perform the experimental procedures described above using this new cell line to determine the mechanism by which C. arvensis extract inhibits angiogenesis.