Taina Matheson and Dr. Leigh Johnson, Integrative Biology
Many flowering plants possess small epidermal hairs and glands. The variation in these features has been used to infer relationships among plant groups. The Phlox family (Polemoniaceae) is one such group. Many species in this family are described as having “woolly or arachnose hairs.” These hairs are present in many taxa that are considered to be unrelated, however, and may not be structurally identical. Thus, it may be unrealistic to score these features the same in phylogenetic analyses. First, we need to accurately describe the variation that is present. The genus Gilia has historically been used as a sort of “other” category within Polemoniaceae. In the past few decades, researchers have attempted to make this genus a more natural group. The relationships of Gilia sect. Kelloggia, a group of three species (5 taxa including subspecies), are in dispute. Grant and Day (1998) placed these species in the genus Allophyllum on the basis of purported similarity in gland morphology. Porter and Johnson (2000) placed the group in the genus Navarrettia. Their findings were based primarily on DNA sequence evidence, but the authors also justified their conclusions on the basis of morphological similarities they believed to exist. Neither group presented convincing morphological evidence.
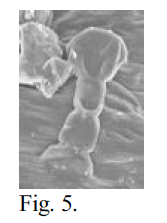
I used scanning electron microscopy (SEM) to examine the epidermal hairs and glands of these species at high magnification, hoping to accurately describe their structure at the cellular level. My objective was to make a comprehensive survey of Navarrettia, Gilia sect. Kelloggia, and Allophyllum, in order to provide a useful description of the variations in gland morphology, and thus help to elucidate the relationships of the groups. Plant material was collected in the field by Dr. Leigh Johnson. Pressed, dehydrated herbarium material was examined for all taxa. Individual flower calyces were dissected out and mounted on aluminum stubs using double-stick tape, then sputter coated with gold. In some cases, brief rehydration was necessary to soften the material so that it would not shatter upon dissection. For some taxa, material had been placed when fresh in 70% FAA, a biological fixative and preservative. This material was prepared by staining with OsO4 and critical point drying, followed by mounting and coating. I examined the prepared specimens using the JEOL 840A SEM in the Integrative Biology microscopy lab. Photomicrographs were captured digitally. Unfortunately, SEM was unable to show the cellular structure of the glandular hairs. In dehydrated material, the general shape of the structures was discernible. The material was often shattered, however, with many of the glandular caps sheared off or severely shrunken. The material preserved in FAA and critical point dried was better, with less shrinkage and breakage. However, the cellular arrangement of the glandular cap still was not visible in most cases. Figures 5 and 6 below show the difference between dehydrated and critical point dried samples of the same species.
We also viewed a few rehydrated samples using the Environmental SEM (ESEM), in which the gaseous composition and water content of the atmosphere in the sample chamber can be controlled. However, dessication of the sample was almost unavoidable, and a water film on the sample surface obscured the features that we wanted to see. Previously, many of the taxa were examined using light microscopy. Rehydrated material was viewed with a high-powered dissecting microscope, and line drawings of the calyx trichomes were made (Johnson unpublished). By using SEM, we hoped to avoid the problem of transparency, which makes it difficult to determine the number and boundaries of cells in the glandular caps. Unfortunately, we could not see the cell boundaries at all in most cases. The figures below compare drawings made from light micrographs to the SEM photomicrographs.
 |
 |
 |
 |
 |
 |
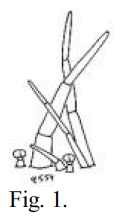
Fig. 1 – A drawing of calyx trichomes of Navarrettia intertexta, made by Leigh Johnson.
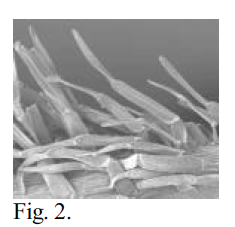
Fig. 2 – SEM photomicrograph of calyx trichomes of N. intertexta at 100x magnification. Dehydrated material. 15 mm WD, 10 KV, 6×10-10 A. Note similarity to Fig. 1.
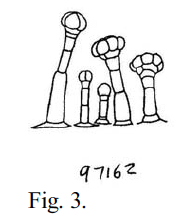
Fig. 3 – A drawing of calyx trichomes of Navarrettia capillaris (Gilia capillaris), made by Leigh Johnson.
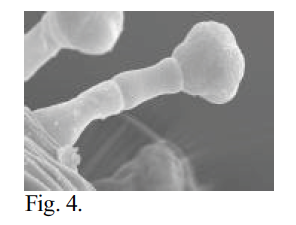
Fig. 4 – SEM photomicrograph of calyx trichomes of N. capillaris at 1200x magnification. Critical point dried material. 15 mm WD, 5 KV, 6×10-11 A. Note lobing of cap suggesting fourcelled arrangement. Compare Fig. 3.
Fig. 5 – SEM photomicrograph of a calyx trichome of N. capillaris at 1000x magnification. Dehydrated material. 15 mm WD, 15 KV, 6×10-11 A. Compare Fig. 4.
Fig. 6 – SEM photomicrograph of a calyx trichome of N. sinistra ssp. sinistra genotype “D” at 1300x magnification. Critical point dried material. 15 mm WD, 5 KV, 6×10-11 A. Possibly the best image obtained in terms of showing the cellular structure of the trichome.
References
- Grant, V. 1998. Primary classification and phylogeny of Polemoniaceae, with comments on molecular cladistics. Amer. J. Bot. 85: 741-752.
- Porter, J.M. and L.A. Johnson. 2000. A phylogenetic classification of Polemoniaceae. Aliso 19: 55-91.
