Janet L Hart and Dr. Kim L O’Neill, Microbiology and Molecular Biology
Non-steroidal anti-inflammatory drugs (NSAIDs) such as sulindac and acetylsalicylic acid (ASA) are frequently used to relieve pain, clotting, fever and inflammation. Recently, NSAIDs have been shown to decrease the occurrence of colorectal cancer and inhibit mammary cancer in rats. To elucidate this mechanism, further research has been done to show an inhibition of matrix metalloproteinases (MMPs) by NSAIDs in lung and prostate carcinomas. Many current chemotherapeutic agents such as doxorubicin and methotrexate (DOX and MTX respectively) rely on the Fas/FasL (CD95) pathway to induce cell death. Because MMPs are able to cleave membrane-bound Fas, these chemotherapeutics are less-effective.
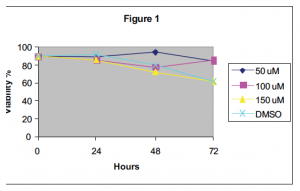
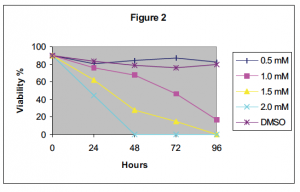
In this study, I treated human acute T lymphocytes concurrently with a NSAID and a Fasdependent chemotherapeutic. The highest concentration of sulindac and ASA that did not induce cell death in Jurkat cells was used (see fig. 1 and 2 respectively). 1 ìM doxorubicin and 10 ìM methotrexate were used to induce cell death. Using flow cytometry, the percent of apoptotic cells was assessed by phosphatidylserine (PS) externalization (ANNEXIN V binding), which is characteristic of cells undergoing apoptosis.
The apoptotic percentage was tested at various timepoints for each of the drugs. Control cells were compared to the cells receiving the chemotherapeutic alone and those receiving the chemotherapeutic + NSAID treatment.
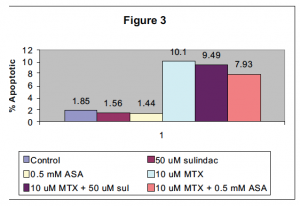
As seen in figure 3, the concurrent treatment of the either ASA or sulindac with methotrexate after 48 hours did not show a significant increase in apoptotic percentage in Jurkat cells.
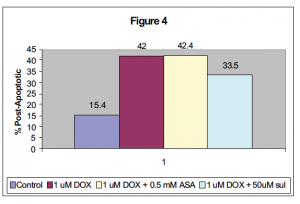
When using doxorubicin, similar results were seen. Figure 4 shows the relative post-apoptotic percentage of cells after a 12 hour treatment with either ASA or sulindac and doxorubicin in Jurkat cells.
Because the flow cytometric results were inconclusive, Western Blots were performed on cell extracts from treated cells to see if there was an inhibition of MMP protein in the cells. Jurkat cells were treated for 24 hours with either 150 ìM sulindac or 1.0 mM ASA. Cell extracts were isolated and protein concentration was equilibrated using the Bradford method. Monoclonal antibodies to MMP-2 and MMP-7 were used. The results from the Western Blot were inconclusive (data not shown).
Although the results do not support my hypothesis, valuable insights were gained from this project. The clinical significance of this research and other related research in combinatorial drug regimes makes this project worthwhile. Further research will be conducted using different drugs, concentrations and cell lines.
 |
 |
 |
 |
Sources:
- Kargmann, S., Charleson, S., Cartwright, M., Frank, J., Mancini, J., Evans, J. and O’Neill, G. (1996) Gastroenterology 111. 445-454.
- Coyne, D.W., Nickols, M., Bertrand, W. and Morrison, A.R. (1992) Am. J. Physiol. 263, F97-F102.
- Jones, D.A., Carleton, D.P., McIntyre, T.M., Zimmerman, G.A. and Prescott, S.M. (1993) J. Biol. Chem. 268, 9054-9094.
- Hamanski, Y., Kitzler, J., Hardman, R., Nettesheim, P. and Eling, T.E. (1993) Arch. Biochem. Biophys. 304, 226-234.
- DuBois, R.N., Tsujii, M., Bishop, P., Awad, J.A., Makita, K. and Lanahan, A. (1994) Am. J. Physiol. 266, G822-G827.
- Hidalgo, M. and Eckhardt, S.G. (2001) J. Natl. Cancer Inst. 93, 178-193.
- Rha, S.Y., Yang, W.I., Kim, J.H., Roh, J.L., Min, J.S. and Lee, K.S. (1998) Oncol. Rep. 5, 875-879.
- Gridley, G., McLaughlin, J.K., Ekbom, A., Klareskog, L., Adami, H.O., Hacker, D.G., Hoover, R. and Fraumeni, J.F., jr. (1993) J. Natl. Cancer Inst. 85, 307-311.
- Williams, C.S., Mann, M. and DuBois, R.N. (1999) Oncogene. 18, 7908-7916.
- Pan, M.R., Chuang, L.Y. and Hung, W.C. (2001) FEBS Lett. 508, 365-368.
- Attiga, F.A., Fernandez, P.M., Weeraratna, A.T., Manyak, M.J. and Patierno, S.R. (2000) Cancer Res. 60, 4629-4637.
- 12. Friese, C., Herr, I., Krammer, P.H. and Debatin, K.M. (1996) Nat. Med. 2, 574-577.
- 13. Mitsiades, N., Yu, W.H., Poulaki, V., Tsokos, M. and Stamenkovic, I. (2001) Cancer Res. 61, 577-581.
