Jon Ahlstrom and Dr. Kim O’Neill, Microbiology
Cancer is the cause of death of 1 in 3 citizens in the United States. A diet rich in fruits and vegetables can reduce cancer deaths by 33%. Cancer prevention from dietary plants may in part be derived from individual phytochemicals inherent to the plant. The identification of plant phytochemicals that have anti-cancer properties and their mode of action is an important area of cancer research.
(-) Epigallocatechin gallate (EGCG) has been shown to prevent cancer in rodents. EGCG is the most abundant photochemical in green tea. The consumption of large amounts of tea has been shown to decrease the risk of several types of cancer and tea is recognized as a cancer preventative in Japan i. There are several postulated mechanisms of molecular prevention of EGCG. One way in which EGCG has been shown to prevent cancer is by altering cellular enzyme activity.
Most chemical agents that cause cancer require transformation by enzymes to become cancerous compounds. The science of mammalian enzymes changing foreign compounds is called “xenobiotics.” There are two types of xenobiotic enzymes: phase I and phase II enzymes. Phase I enzymes are so classified because they can change foreign chemicals into reactive, cancer causing compounds (carcinogens) whereas phase II enzymes can neutralize the reactive compounds. The balance of phase I and phase II enzyme activity in the body determines if cancer results or not. Two ways in which EGCG could prevent cancer is to decrease the activity of the activating phase I enzymes or to increase the neutralizing phase II enzyme activity. In the protection of rodents against the chemical benzo[a]pyrene (B[a]P), EGCG has been shown to both decrease phase I activity and increase phase II activity.
My proposal for the ORCA was to develop assays for the detection of phase I and phase II enzyme activity in human HepG2 cells. The glutathione-S-transferase assay (GST) is based on the spectrophotometric color change of when the enzyme glutathione-S-transferase conjugates glutathione to the substrate 1-chloro-2,4-dinitrobenzene. The 7-ethoxyresorufin-O-dealkylation assay (EROD) is specific for the phase I enzyme cytochrome P450 1A. The EROD is a spectrofluorometric assay which detects for the conversion of 7-ethoxyresorufin to resorufin. Both enzymes GST and cytochrome P450 1A have been shown to be important for the detoxification and activation of B[a]P respectively. HepG2 cells were used to study B[a]P activation and detoxification. The Alkaline Comet Assay, which can quantify DNA strand breaks on the single cell level, was the technique used to detect B[a]P DNA damage.
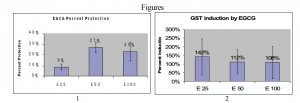
EGCG protected against B[a]P DNA damage in HepG2 cells as shown by the Alkaline Comet Assay (see FIG 1). The enzyme assays were then performed on HepG2 cells treated in the same way. One of the difficulties of this project was the best way to isolate the enzymes. Gutathione- S-transferase is a cytosolic protein and cytochrome P450 1A is membrane bound protein in the smooth endoplasmic reticulum. Some options were to freeze-thaw the cells several times, to use a glass tube with a tight fitting plunger called a Douse homogenizer, or to use ultrasound to disrupt cell membranes. Freeze thaw has been shown to partially inactivate cytochrome P450 enzymes and using a Douse homogenizer is cumbersome for multiple samples—hence sonification was chosen. One success of this project was a reliable method of disrupting cell membranes to harvest the enzymes.
Numerous protocols exist for the GST and EROD assays and discovering the best one also took much time. A protocol that adapts the GST assay to a 96 well plate was found ii, allowing several experiments to be processed at once. The results of the GST assay for EGCG are shown in FIG 2. EGCG treatment against B[a]P increased GST activity on average. Therefore, one of the mechanisms of EGCG protection against B[a]P may be to increase phase II enzyme activity. A simple protocol for the EROD was attempted iii, but as yet no EROD results have been obtained. When the cofactor NADPH is added, there is an immediate reduction in fluorescent intensity of 7-ethozxy resorufin. The product resorufin should then be formed in 5 min—and it is not. There are several possibilities why the product is not formed even though the metabolite disappears. However, it is still not known why the EROD does not seem to work even after months of effort. I have now graduated from BYU and others will continue working on the EROD assay.
In conclusion, EGCG has an apparently protective affect against the suspected carcinogen B[a]P in human HepG2 cells. One mode of protection may be by the induction of the phase II enzyme glutathione-S-transferase as shown by the GST assay.
Figure 1: HepG2 cells treated simultanesouly with 25 μM B[a]P and 25, 50, or 100 μM EGCG for 24 hrs. The data represent the average percent protection as compared to B[a]P alone from three separate studies. The error bars show the standard deviation.
Figure 2: GST activity in enzymes homogenates from HepG2 cells treated the same as in Figure 1. The data show the average percent increase in GST activity as compared to an untreated control (100%) from five separate studies. The error bars show the standard deviation.
________________________________________
i H. Fujiki, et al. Cancer inhibition by green tea, Mutation Res. 402 (1998) 307-310.
ii S. Sharma, et al. Identification of chemopreventive agents by screening for induction of glutathione-S-transferase as a biomarker, Methods Cell Sci. 19 (1997) 49-52.
iii T. Chang and D. Waxman. Enzymatic anaylsis of cDNA-expressed human CYP1A1, CYP1A2, and CYP1B1 with 7-ethoxyresorufin as substrate, pps103-109. In: Cytochrome P450 protocols, I. Phillips and E. Shephard eds., Humana press, Totowa, 1984.