Allison Sparks and Dr. William McCleary, Microbiology
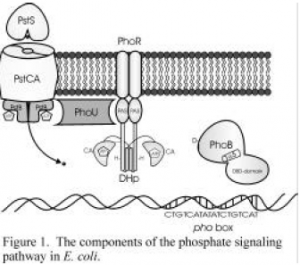
All bacteria must have a way to control cell functions when their outside environment changes. E.coli uses a phosphate signaling pathway to accomplish this. In the bacterial cell membrane, there are proteins that take up phosphate from the environment, transports it into the cell, and triggers transcription of certain genes. The system is comprised of an ATP-dependent transport system which consists of a periplasmic substrate binding protein, PstS, three membrane proteins, PstC, PstA, and PstB, a regulator, PhoU, a transmembrane histidine kinase, PhoR, and a response regulator PhoB. In this system, the response regulator, PhoB is phosphorlated and then activates transcription of Pho regulon genes. PhoB must undergo a conformational change after being phosporalated which makes it active. It is the purpose of this project to investigate these conformational changes by mapping changes in protease accessibility upon PhoB phosphorylation.
The phosphate signaling pathway is widespread in the prokaryotic realm, but there are still details which have not been elucidated. These signal transduction pathways that involve protein phosporylation are a common theme in prokaryotic and eukaryotic systems. These systems control development, differentiation, growth, DNA replication, cell division and other adaptive responses. By understanding PhoB, we can understand the general mechanisms involved in protein phosphorylation and the effects of phosphorylation on protein activity and structure. Information obtained from this research will be very valuable in understanding how phosphorylation modulates the activity of PhoB and other related systems. The proposed research also has gravity in the area of antimicrobial agents. The phosphate signaling pathway is a type of two-component system, which is found in many disease causing agents. If the conformational changes of PhoB are fully understood, it could lead to the design of specific inhibitors, which could be used as antimicrobial agents to cure disease.
We hypothesize that PhoB undergoes conformational changes when it is phosphoryated. We plan to prove this hypothesis by doing protease sensitivity experiments on phosphorylated and unphosphorylated PhoB.
At this time, we are unable to determine the conformational change of PhoB. We were able to cut the protein with trypsin and see different size protein bands in the SDS page, but the bands were not defined enough to tell a difference in the 20 min. incubation and the 120 min. incubation. Many adjustments were made in the concentration of trypsin and incubation times to produce more viable data, but time ran out before we could get a working gel with the desired data. Due to Often the gels would not set up for any apparent reason. There were also many times were the equipment, whi have the appropriate bands. The project was very frustrating and due the nature of the experiment very time consuming.
The project will be taken up by another student and ho soon me discovered. I found the project to be very educational and it gave me great experience in the laboratory.
References
- Makino, K., H. Shinagawa, M. Amemura, and A. Nakata. 1986. Nucleotide sequence of the p gene, the positive regulatory gene for the phosphate regulon of Echerichia coli -12. J 190: 37 44
- McCleary,W.R., and D.R. Zusman. 1990. Purification and characterization of the Myxococcus FrzE protein shows that it has autophophorylation activity. J. Bacteriol. : 6661- M. Leid.(2002)Mass-spectometric analysis of agonist-induced retinoic acid receptor ( conformational change. Biochem. J. 362: 173-181.
