Peter Crane and Dr. Robert Seegmiller, Physiology and Developmental Biology
Normal articular cartilage is characterized by a resilient collagen and proteoglycan matrix interdispersed with chondrocytes. Collagen type II is the most abundant collagen found in the matrix and is responsible for providing tensile strength to the tissue. The highly conserved Col2a1 gene is responsible for coding all type II collagen á-helix chains.1 The process of enzymatic MMP cleavage is a natural part of collagen recycling, but has been observed to be upregulated in articular cartilage of osteoarthritis (OA) patients. It is hypothesized that further denaturation (unwinding) of enzymatically cleaved collagen takes place following enzymatic cleavage by MMP. Because increased degraded type II collagen has been observed in human OA patients through use of immunohistchemistry, there is a need for an animal model to study and use to develop treatments.2, 3 C3H mice with disproportionate micromelia (Dmm) carry a radiation-induced mutation in the region of the Col2a1 gene that codes the C-propeptide of type II collagen.
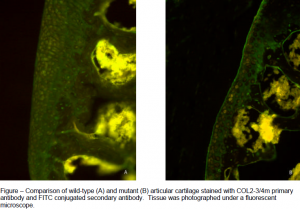
The purpose of this study was to test the utility of the Dmm mouse as an animal model to study the pathogenesis of OA. Dmm is the only known homozygous Col2a1 Cpropeptide mutation animal model.4 The type II collagen specific antibody COL2-3/4m was used to immunolocalize degraded type II collagen in Dmm mice and verify their utility as an animal model for studying degraded type II collagen associated with OA.5 Knee joints from 3, 6, 9, and 12 month-old control (+/+) and heterozygous (D/+) animals were used in the present study. The knee joints were fixed in 4% formaldehyde and then decalcified in formic acid decalcifier. Next, the joints were sectioned coronally at 6 mm using a cryostat and mounted on slides. After preparative blocking steps the sections were incubated overnight at 4° C with COL2-3/4m primary antibody. To visualize the staining the sections were then incubated with a secondary antibody conjugated with a fluorescing chromophore (FITC) and observed and photographed under a fluorescent microscope. After photographing, pixels stained at the superficial zone of the articular cartilage were quantified using the Adobe Photoshop 7.0 magic wand function. A nonparametric analysis of variance using ranks of the quantified values was performed using SAS software.
Dmm heterozygotes showed more staining for degraded collagen than wild-type mice of the same age. Differences between mutant and control degraded collagen were significant (P<0.05) in six-, nine-, and twelve-month animals. The three-month animals showed no significant differences between wild type and mutant degraded collagen (P=0.34) and staining did not differ significantly between age groups. The quantified values demonstrate that the Dmm heterozygotes develop heightened levels of degraded collagen near six months of age and that the levels remain heightened at approximately the same level until at least twelve months.
The present study suggests the utility of Dmm heterozygotes as an appropriate model for the study of degraded type II collagen associated with genetically predisposed osteoarthritis. The data presented herein will be submitted for publication in Osteoarthritis and Cartilage. Future studies using Dmm mice might include developing and testing therapeutic interventions targeted at reducing collagenase activity, regulating interleukin levels related to collagen degradation, or preparing gene therapy treatments for type II collagen C-propeptide defects.6
References
- Buckwalter JA, Mankin HJ. Articular Cartilage: Tissue Design and Chondrocyte-Matrix Interactions. J Bone Joint Surg Am 1997;79-A:600-11.
- Billinghurst RC, Dahlberg L, Ionescu M, Reiner A, Bourne R, Rorabeck C, et al. Enhanced cleavage of type II collagen by collagenases in osteoarthritic articular cartilage. J Clin Invest 1997;99:1534-45.
- Hyttinen MM, Toyras J, Lapvetelainen T, Lindblom J, Prockop DJ, Li SW, et al. Inactivation of one allele of the type II collagen gene alters the collagen network in murine articular cartilage and makes cartilage softer. Ann Rheum Dis 2001;60:262-8.
- Pace JM, Li Y, Seegmiller RE, Teuscher C, Taylor BA, Olsen BR. Disproportionate micromelia (Dmm) in mice caused by a mutation in the C-propeptide coding region of Col2a1. Dev Dyn 1997;208:25-33.
- Hollander AP, Heathfield TF, Webber C, Iwata Y, Bourne R, Rorabeck C, et al. Increased damage to type II collagen in osteoarthritic articular cartilage detected by a new immunoassay. J Clin Invest 1994;93:1722-32.
- Acknowledgements: I thank Dr. A. Robin Poole of the Joint Diseases Laboratory, Shriners Hospital for Children in Montreal, Canada for generously providing the COL2-3/4m antibody and Dr. Dennis Eggett of the Center for Statistical Consultation and Collaborative Research at Brigham Young University for performing the statistical analyses.