Deborah Adams and Dr. Dixon Woodbury, Physiology and Developmental Biology
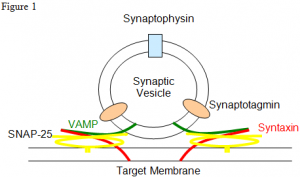
SNARE proteins are involved in transporting molecules within our cells, but their mechanism of action is not well known (Figure 1). It is established that SNARE proteins aid in the process of vesicles docking at target membranes. When neuronal cells are excited calcium enters the cell and the docked vesicles fuse to the target membrane releasing their contents into the intercellular space called the synapse. It is not exactly known by what method calcium induces membrane fusion but it has been shown that syntaxin and synaptobrevin, or VAMP, are minimal machinery for membrane fusion (1).
SNARE proteins localize in membranes, so part of the protein structure is hydrophobic. This property makes these proteins very difficult to work with because the hydrophobic domain tends to non-specifically interact with many substances. It is possible that some of the SNARE protein interactions that have been reported are due to non-specific binding of the hydrophobic regions. I looked very carefully at the binding properties of SNARE proteins to determine whether the interactions were due to specific or non-specific binding. This was accomplished partly by using full-length proteins and truncated versions that are missing the hydrophobic membrane spanning region.
To investigate binding properties a standard binding assay, that determined whether interactions between SNARE proteins and other components occur. One goal was to reconstitute VAMP into artificial vesicles and study what proteins bound to it, but instead glutathione agarose beads were used in these binding assays because of convenience. It was easy to obtain proteins attached to these beads because of the purification process used. VAMP, syntaxin, or SNAP-25 was attached to these beads and incubated with other proteins. The beads were then washed and assayed for the presence of other proteins. My results confirmed that syntaxin, SNAP-25 and VAMP all bind to each other in a specific manner. Another goal of this project was to investigate the effect of regulators like munc18. Unfortunately I was not able to accomplish this goal because the protein did not become available to me in time to perform these experiments.
After the binding assays were performed, function assays were run to determine the effect of binding on membrane fusion. As mention previously it has been shown that syntaxin and VAMP are the minimal machinery for membrane fusion (1). Those experiments were done using the nystatin/ ergosterol fusion assay. This is the same assay that I used in my experiments. VAMP was reconstituted into artificial vesicles, while syntaxin, and/or SNAP-25 were reconstituted into an artificial membrane. Fusion of the vesicles to the membrane was detected by a sharp increase in conductance, which then decayed away. In the first set of experiments done, VAMP vesicles were used and syntaxin or syntaxin/SNAP-25 was reconstituted into the membrane. Results showed that adding SNAP-25 enhanced membrane fusion. This experiment was also done with a mutated form of syntaxin in which one amino acid was changed to a negative residue that mimicked a phosphorylated state. It was predicted that this mutation would possibly increase fusion rates. This data showed that when only mutated syntaxin was in the membrane there was more fusion than with wild type syntaxin. SNAP-25 again increased fusion rates, but not as dramatically as with the wild type.
The next step in my research was to try and repeat some of these results, because the functional assay utilized in these experiments is highly variable. At the same time another goal was to compare the fusion rates with wild type syntaxin/SNAP-25 and mutated syntaxin/SNAP-25. Another aim of these experiments was to perform many trails and obtain a clear result on which combination of protein led to more fusion. Unfortunately I began these trials, but was unable to finish as many as I would have hoped and the results obtain so far are not very clear. All experiments had high fusion rates which made it difficult to count all of the fusion and thus tell which proteins led to more. Analysis did show that the mutated syntaxin supported a slightly higher fusion rate.
In the future these results will be repeated to obtain cleared results. Other protein will also be utilized such as munc18 and synaptotagmin. The effects of phosphorylation will also be studied with syntaxin as well as the other SNARE proteins.
References:
- Woodbury, D.J., Rognlien, K. The t-SNARE syntaxin is sufficient for spontaneous fusion of synaptic vesicles to planar membranes. Cell Biology International. 24:809-818 (2000).
