Michael Bingham and Dr. Jeff Bullock
Abstract
The principle objectives of this research analysis were to determine the uptake and turnover rates of carbon and nitrogen stable isotopes in fish and to establish if this isotope verification procedure is possible by testing tissue samples of their fins. This investigation is separated into two main experiments: first, the stable isotope incorporation in the new growth of fin tissue, and second, the rate of stable isotope turnover in the old growth of fin tissue. Both experiments of this study were done with Gambusia affinis, a species of fish better know as mosquito fish.
The first experiment includes 36 fish analyzed at 3 and 5 weeks. The fish in this experiment had their caudal fins clipped at the beginning of the experiment (week 0), which allowed for stable isotope incorporation into the new fin tissue growth.
The second experiment involves 36 fish analyzed at 2 and 4 weeks, which allowed the rate of stable isotope turnover in old growth fin tissue to be verified.
Both experiments divided the fish into three groups, and each group was fed a different food over the duration of the experiment. By feeding the fish these assigned foods, the researchers were able to introduce different isotope ratios into the fishes’ diet.
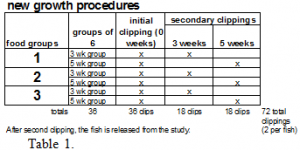
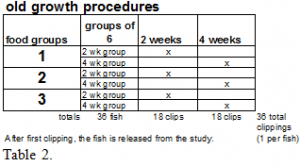
-See tables 1 & 2—Procedure tables.
The isotope ratios of the samples were analyzed using an isotope ratio mass spectrometer.
We were surprised to find that stable isotope turnover began to manifest in the old growth experiment as quickly as two weeks. Isotope ratios for each group continued to converge in the fourth week toward each respective food isotope ratio. The results for the new growth were even more dramatic.
INTRODUCTION
The principle objectives of this study are to determine the uptake and turnover rates of 13C and 15N stable isotopes in fish and to establish if this isotope verification procedure is possible by testing fin tissue samples. Some researchers have begun using fin tissue in their stable isotope studies but little has been done to verify the accuracy of this method (Charles, Roussel, & Cunjak 2004).
Stable isotope ratios have been used successfully for the last decade to determine dietary sources, construct food webs, and diagram migratory patterns.
Many elements occur in two or more isotopic forms. The stable (non-radioactive) isotope forms of an element differ only in the number of neutrons but possess virtually identical chemical properties. For example, carbon and nitrogen most commonly occur as 12C and 14N but are also found as 13C and 15N, each having one extra neutron.
Plants and animals possess unique ratios of light and heavy isotopes depending largely on their diet. Carbon is found in carbohydrates which are used either for energy or stored as fat tissue. Both carbon and nitrogen are found in protein which can be used either for energy or for biological building material and, thus, be incorporated into the consumer’s body tissues. Therefore, the ratios of stable isotopes found in animal tissue reflect, to a certain extent, the animal’s diet.
In fine, we will carry out a study in order to determine the rate of turnover of stable carbon and nitrogen isotopes using clippings of fish caudal fin tissue.
Typically, to obtain tissue samples for stable isotope analysis, the fish is sacrificed and muscle or organ tissue is used to perform the analysis. Fin tissue, on the other hand, is quickly regenerated in most species. The use of fin tissue would be a novel approach to biological stable isotope research. Calls have even been made to validate the use of fin tissue in this manner (McCarthy & Waldron 2000). We did find one test in which isotope ratios of fin samples were compared to those of muscle plugs, but this was done as a side note and was not pursued as a main objective (Shannon, Blinn, Haden, Benenati, & Wilson 2001).
Understanding food webs is a critical step in the development of effective wildlife conservation. Stable isotope research has become an invaluable tool in constructing these webs.
However, killing the fish to collect muscle and organ tissue is not an option when studying endangered species. Using fin clippings would be a harmless and convenient method for obtaining tissue samples for isotope analysis. With this method, the diet of endangered fish species, some of which are found only in Utah, could be studied; seasonal changes in diet might even be monitored.
This study is separated into two main experiments: Experiment 1—stable isotope incorporation in new growth fin tissue, and Experiment 2—rate of stable isotope turnover in old growth fin tissue.
In Experiment 1, the level of incorporation into new fin tissue of stable isotopes found in the fishes’ diet will be determined. This experiment will consist of three groups of fish consisting of 12 fish each (each group defined by the diet administered). All 36 fish in this experiment will begin the test by having their caudal fins clipped; the 13C/12C and 15N/14N ratios of the fin clippings will then be determined. The caudal fin of the mosquito fish will be clipped to no closer than 1 mm from the base of the fin rays.
After the initial fin clipping, a diet unique (see diet description below) to each group will be administered to the fish for up to five weeks. The 13C/12C and 15N/14N stable isotope ratios of each diet are previously determined. By feeding each group isotopically different diets, variations in the stable isotope ratios are introduced.
After the initial fin clipping, the caudal fin of each fish will be clipped once more. Six fish from each group will be clipped at three weeks and six more from each at five weeks. Each fish will be released from the study after the second clipping.
In our pilot study, mosquito fish were found to completely re-grow their caudal fin in less than three weeks. In searching the literature, we found that zebra fish can re-grow 65% of their caudle fin in just 7 days (Zodrow J. M., Tanguay R. L. 2003). Zebra fish may also be a suitable species for similar studies.
Sample sizes at each interval will be six fish from each group, thus a total of 54 fish in this test (New Growth Test). Thus, we will be able to measure the degree to which the new isotopic signal is incorporated into the fin from the diet.
In Experiment 2, the rate of turnover of stable isotopes in old-growth fin tissue caused by diet change will be determined. This experiment will also consist of three groups. These three groups are separate from the new growth experiment and will be comprised of 36 fish each (groups again defined by the diet administered). Each group will be fed on one of the diets in the new growth study.
Caudal fins in this experiment will be harvested and analyzed from each group at two and six weeks (six fins from each group at each time interval). In this study each fish will only be clipped once and then released from the study.
Sample sizes at each interval will be six fish from each group, thus a total of 36 fish in this test (old growth test). With this method, we hope to be able to measure the rate of stable isotope turnover in old fin tissue.
The following protocol has been established in the case of multiple deaths or illness in fish subjects:
• If more than two fish die in one of the sub-groups of six, the remaining fish from that subgroup will be dismissed from the test.
• If such deaths repeatedly occur at a biased rate in one of the large groups (defined by unique diet), that group will be temporarily dismissed from the test and may be used in a future, repeat test under a different diet.
• If illness or reduced activity becomes apparent in one of the large groups (defined by unique diet), the diet will be reevaluated and, if necessary, the group will be temporarily dismissed from the test.
• If repeated deaths occur throughout all test groups causing dismissal under the above criteria, testing will be stopped and the procedures will be reevaluated. If the procedures can be acceptably amended, testing will begin anew under the new methods.
MATERIALS AND METHODS
Materials
The following materials were used in this study:
• Gabusia affinis , mosquito fish (72)
• Ziploc 400 ml plastic containers with perforated lids (72)
• Environment control chambers (2)
• Fish foods (3 varieties)
Group 1: Penn-Plax Tropical Fish Food Diet
Ingredients: First Quality Fish Meal, Soya, Wheat Gluten, Wheat Germ Meal, Prawn Meal, Dried Yeast, Fish Oil, Lecithin, Spirulina, Multi Vitamins, Essential Trace Elements and Multiple Amino Acid Additive.
Group 2: Wardley Goldfish Crumbles
Ingredients: Ground Corn, Fish Meal, Soy Flour, Corn Meal, Wheat Germ Meal, Dehydrated Alfalfa Meal, Brewers Dried Yeast, Ground Whole Wheat, Shrimp Meal, Fish Oil, Natural Flavor Fish Protein Concentrate, L-Ascrbyl-2-Polyphosphate, Vitamin Premix Containing (Wheat Middlings, Vitamin A Supplement, Vitamin D3 Supplement, Vitamin E Supplement, Vitamin B12 Supplement, Riboflavin Supplement, Niacin Supplement, Calcium Panthotenate, Menadione Sodium Bisulfite Complex, Folic Acid, Pyridoxine HCl, Thiamine Mononitrate, d-Biotin), Choline Chloride, betain, Marigold Petal Extract, Ethoxyquin.
Group 3: HBH Veggie Wafers
Ingredients: Spirulina Powder, Alfalfa Meal, Soy Flour, Wheat Flour, Wheat Gluten, Wheat Germ, Brown Rice Flour, Wheat Starch, Spinach Powder, Carrot Powder, Stabilized Fish Oil, Brewer’s Dry Yeast, Dry Lecithin, Monocalcium Phosphate, Zucchini Flakes, L-ascorbyl-2-polyphosphate (source of Vitamin C), Vitamin A Acetate, Choline Chloride, a-Tocopheryl Acetate (source of Vitamin E), Niacin, d-Calcium Pantothenate, Riboflavin, Thiamin, Biotin, Pyridoxine HCl, Folic Acid, D-Activated Animal Sterol (source of Vitamin D3), Vitamin B12 Supplement, Zinc Proteinate, Copper Proteinate, Manganese Proteinate, Iron Proteinate, Ethoxyquin.
Note: These foods were fed for one month to small groups of mosquito fish in our pilot test without any noticeable difference in activity or health among the groups.
The above foods were selected because of the range they represent in carbon and nitrogen stable isotope ratios.
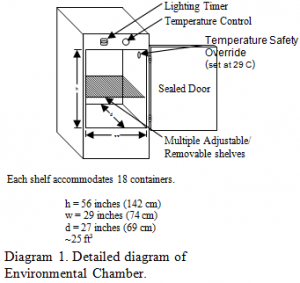
To establish the appropriate, controlled environmental conditions for the research the following was done:
1. Prepare 20 gallons of aged water by aerating for at least 72 hours.
2. Fill each Ziploc™ 400 ml plastic containers with aged water and put on the perforated lids.
3. Set the environmental chamber controls to allow for temperature settings of 23.5 C and the lighting on a 12:12, normal lighting cycle.
Methods
Note: Because no data relevant to our experiment was available on mosquito fish, we conducted a small pilot experiment in order to develop our procedures.
We obtained ten mosquito fish from Dr. Mark Belk of Brigham Young University. In this pilot study, we observed rate of fin regeneration, dry weight of caudal fin clipping (0.5 mg is required for the mass spectrometer), tolerance to the foods particular to the mosquito fish study, water change preference, and compatibility with the 400 ml plastic containers.
We found that the caudal fin, when clipped to within 1 mm of base of the fin rays, will re-grow completely within three weeks with little noticeable difference in rate of re-growth among the three different diets. The dry weight of the caudal fin, from the tips of the rays to 1 mm from the base of the rays, is just over 0.5 mg.
We aged the water in glass tanks with aerators to neutralize chlorine in the tap water and to remove any ammonia that may have been present. At the end of one week, when kept in the 400 ml containers under these conditions, the fish appeared active and the water unclouded.
Based on this, we decided that changing the water once a week without aeration would be sufficiently frequent for the mosquito fish, which are often found thriving in brackish, slow, or standing streams. We held these fish for one month on the separate diets; at the end of the month all fish remained equally active and apparently healthy.
We also developed methods for restraining and clipping. The individual fish is placed in a large weigh boat with water; this restricts the swimming area. Sufficiently small fish are coaxed to swim into the large end of a Pasteur pipette, and the exposed tail is clipped (time in the pipette is no longer than 20 seconds). Larger fish are gently restrained with finger tips in the water (handling time with this method is about 10 seconds). All fish survived these procedures and completed the one month pilot test.
In order to predict the number of samples required for statistical significance, a power analysis based upon values available in the literature (Sotiropoulos M. A., Tonn W. M., Wassenaar L.I. 2004) shows that a sample size of six fish per group would have a power in excess of 0.9 to distinguish differences between groups of 1 per mil or more at P
Experiment I
The 36 mosquito fish were divided up into three groups: Test Group I, Test Group II, and Test Group III. Twelve fish were placed into each group and their caudal fins were harvested to establish initial stable isotope ratios.
The preliminary test was conducted to determine the variation of stable carbon and nitrogen (δ13C and δ15N) ratios of the groups using an isotope ratio mass spectrometer (IRMS). Furthermore, the preliminary test included a test of the fish food in order to determine the variation of stable nitrogen (δ15N) and carbon (δ13C) ratios in the food.
Over a five week period, the fish in test groups I-III were fed the various fish foods with the corresponding isotope ratios assigned to their group. Through this method, variations in the stable nitrogen (δ15N) and carbon (δ13C) ratios are introduced.
The fish were placed into the Ziploc™ 400 ml plastic containers with aged water and the perforated lids were secured onto the containers. The fish were fed every Monday, Wednesday, and Friday for the duration of the experiment, and the water in the individual 400 ml containers was exchanged for clean, aged water every Monday.
At the end of the three and five week periods, samples were gathered and tested from each group. Six fish from each group, a total of eighteen fish, had their fins clipped and tested for uptake and turnover levels in the stable-isotope concentration.
A blind study, by an independent tester, was conducted.
The 36 mosquito fish were divided up into three groups: Test Group I, Test Group II, and Test Group III. Twelve fish were placed into each group.
The same guidelines followed in Experiment I for feeding, housing, and caring for the fish were followed in this experiment.
Caudal fins in this experiment were harvested and analyzed from each group at two and four weeks (six fins from each group at each time interval). A blind study by an independent tester was conducted to be able to measure the rate of stable isotope turnover in old fin tissue.
Experiment I & II
In order to minimize the affect of food contamination on the fin samples prior to clipping, the fish were placed in containers with fresh water and no food in order to rinse away any food debris. They were left in this fresh water for at least two hours before the caudal fins were clipped.
The clipped fin samples were dried before being prepared as mass spectrometer samples. Each fin was cut into squares smaller that 1 mm2. A total weight of 0.5-1.0 mg worth of squares was placed in a mass spec sample tin and folded.
Five standards were used as reference material to determine sample repeatability. Results are expressed with standard deviation bars. Isotope ratios are expressed in delta (δ) notation as parts per thousand (per mil, 0/00) differences from a standard as follows:
δ13C or δ15N(0/00) = [(Rsample – Rstandard) / Rstandard] x 1000, where R denotes 13C/12C or 15N/14N. All results are reported relative to Pee Dee Belemnite limestone standard δ13C and to atmospheric nitrogen for δ15N.
RESULTS
In our hypothesis we stated that this study would be able to determine the uptake and turnover rates of stable isotopes in fish and to establish if this isotope verification procedure would be possible by testing fin tissue samples.
Upon evaluating the data, we see a trend for both the nitrogen and carbon ratios to converge towards that of the corresponding food stable isotope ratios. This fact is shown both numerically and graphically, for both the experiments of new growth and old growth.
The graph labeled “New Growth Turnover (Nitrogen Stable Isotope Ratios)” plots the results obtained after three weeks of feeding each group the appropriate food and displays the uptake and turnover rate of the δ15N ratios in Experiment II. Additionally, the δ15N isotope ratios of each food group are plotted; Group I δ15N = 7.240/00, Group II δ15N = 5.230/00, and Group III δ15N = 1.880/00. Groups I, II, III begin with a δ15N value of 14.60/00. The plot shows the converging trend for the three groups to move towards the nitrogen isotope ratios of each corresponding food group; Group I to a δ15N value of 11.120/00, Group II to a δ15N value of 10.310/00, Group III to a value δ15N of 9.140/00.
Note: After running the prepared samples on the IRMS, fin samples harvested for the five week new growth were not accurate due to mechanical problems. In other words the samples tested for nitrogen isotope ratios were not able to be evaluated due to testing errors in the IRMS. Consequently, we did not plot the new growth δ15N from the fins that were harvested at the five week period.
The graph labeled “New Growth Turnover (Carbon Stable Isotope Ratios)” plots the results obtained after five weeks and displays the uptake and turnover rate of the carbon ratios in Experiment I. Additionally, the δ13C isotope ratios of each food group are plotted: Group I δ13C = -20.380/00, Group II delta (13) C = -17.280/00, and Group III δ13C= -25.80/00. The plot shows the converging trend for the three groups to move towards the δ13C isotope ratios of each corresponding food group. At three weeks Group I moves to a δ13C value of -19.590/00, Group II to a δ13C value of -16.540/00, and Group III to a value δ13C of -21.580/00. At five weeks Group I moves to a δ13C value of -18.010/00, Group II to a δ13C value of -16.600/00, and Group III to a value δ13C of -22.680/00.
The graph labeled “Old Growth Turnover (Nitrogen Stable Isotope Ratios)” plots the results obtained after two weeks and four weeks and the uptake and turnover rate of the δ15N ratios in Experiment II. Additionally, the δ15N isotope ratios of each food group are plotted; Group I δ15N = 7.240/00, Group II δ15N = 5.230/00, and Group III δ15N = 1.880/00. Groups I, II, III begin with a δ15N value of 14.570/00. The plot shows a converging trend for the three groups to move towards the δ15N isotope ratios of each corresponding food group. After two weeks Group I changes to a δ15N value of 13.190/00, Groups II and III shift to a δ15N value of 12.850/00. At four weeks Group I changes to a δ15N value of 12.440/00, Group II to a δ15N value of 11.590/00, and Group III shifts to a δ15N value of 10.830/00.
The graph labeled “Old Growth Turnover (Carbon Stable Isotope Ratios)” plots the results obtained after two weeks and four weeks and the uptake and turnover rate of the δ13C ratios in Experiment II. Moreover, the δ13C isotope ratios of each food group are plotted: Group I δ13C = -20.380/00, Group II δ13C = -17.280/00, and Group III δ13C = -25.80/00. Groups I, II, III begin with a δ13C value of -17.760/00. The plot shows a converging trend for the three groups to move towards the δ13C isotope ratios of each corresponding food group. After two weeks Group I changes to a δ13C value of -17.990/00, Group II to a δ13C value of -16.710/00, Group III to a value δ13C of -18.660/00. At five weeks Group I moves to a δ13C value of –18.530/00, Group II to a δ13C value of -16.840/00, and Group III to a value δ13C of –20.950/00.
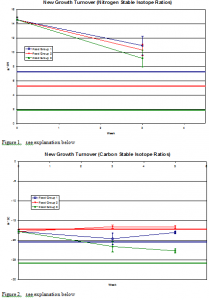
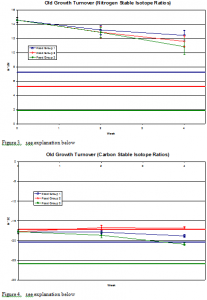
Figures 1-4 Explanation: Comparison of fin and corresponding food isotope ratios. Horizontal lines on the charts represent the stable isotope ratios of the different food groups. The sloped lines represent the stable isotope ratios found in the fish fins. The colors of the lines relate the fins to the diets of the fish (the fins that produced the red sloped line were fed on the food represented by the horizontal red line), and the number of each series, 1, 2, and 3, correspond to these food groups as outlined in the study.
DISCUSSION
The results were statistically significant and are shown graphically with standard deviation bars. It should be mentioned that after running the prepared samples on the IRMS, those fin samples harvested for the five-week new growth experiment encountered evaluation problems. In other words, the nitrogen samples were not able to be evaluated due to mechanical errors in the IRMS, and for this reason we did not plot the findings on the new growth nitrogen graph for the five-week sampling.
In this experiment, we took two groups of mosquito fish and measured the rate of change in isotope ratio affected by diet over a five week process. Our hypothesis was supported in that the stable isotope ratios in the fish fin tissue began to reconcile towards the stable isotope ratios of each fish’s respective diet.
As might be expected, in the new growth experiment the fin tissue isotope ratios changed at a quicker rate than the old growth fish fin samples. This data shows a strong relationship between the length of the experiment and the correlation between nitrogen (δ15N) and carbon (δ13C) ratios in fish and their corresponding diet.
The level of isotope turnover converged towards the stable isotope level for both the old and new growth fishes. For the old growth experiment, the degree of convergence was not as substantial as was the turnover for the new growth.
This observation is supported both statically and graphically. On the graphs shown, one can see that the slope is steeper on the nitrogen new growth samples than on the nitrogen old growth samples. This is significant in showing that new growth tissue samples reflect diet more quickly than old growth tissue samples.
For the overall experiment, we demonstrated that it was possible to determine correlation between diet and tissue samples using the caudal fins in the Gambusia affinis. Previous studies have shown that the ratios of stable isotopes found in animal tissue reflect the animal’s diet to some degree (Gannes, O’Brien, and Del Rio 1996).
Carbon is found in carbohydrates which are used either for energy or stored as fat tissue. Both carbon and nitrogen are found in protein which can be used either for energy or for biological building matter which is found in the consumer’s body tissue.
Traditionally, researchers have obtained the ratios of stable isotopes in animals by sacrificing the animal to obtain tissue samples of their organs or muscle. This study provides basis for the use of fin tissue as an alternative. According to our results, experiments can be successfully conducted on endangered species without further harm to the population. This method would in turn help scientists to know more about the diets of these species. The seasonal changes in diet could also be monitored and the information used in developing better ecological measures.
LITURATURE CITED
- Charles, K., Roussel, J. M., & Cunjak, R. A. 2004. Estimating the contribution of sympatric anadromous and freshwater resident brown trout to juvenile production. Marine and Freshwater Research 55:185-191.
- Gannes, L. Z., O’Brien, D. M., & Del Rio, C. M. 1996. Stable isotopes in animal ecology: assumptions, caveats, and a call for more laboratory experiments. Department of Ecology and Evolutionary Biology, Princeton University.
- McCarthy, I. D., & Waldron, S. 2000. Identifying migratory Salmo trutta using carbon and nitrogen stable isotope ratios. Rapid Communications in Mass Spectrometry 14(15):1325-1331.
- Shannon, J. P., Blinn, D. W., Haden, G. A., Benenati, E. P., & Wilson, K. P. 2001. Food web implications of delta C-13 and delta N-15 variability over 370 km of the regulated Colorado River USA. Isotopes in Environmental and Health Studies 57:179-191.
- Sotriopoulos, M. A., Tonn, W. M., & Wassenaar, L. I. 2004. Effects of fish tissues: potential consequences for food web studies. Ecology of Freshwater Fish 13:155-160.
- Zodrow J. M., & Tanguay, R. L. 2003. 1,2,7,8-tetrachlorodibenzo-p-dioxin inhibits zebra fish caudal fin regeneration. Toxicological Sciences 76(1): 151-161.
