Elizabeth H. Norcross and Dr. William R. McCleary
Every living cell requires a means of sensing its environment and responding appropriately. Even a single cell needs to coordinate its functions to survive. One way cells process environmental stimuli is through signal transduction pathways. Types of signaling used in bacteria include two component systems. “Two component” refers to the two parts of the system, a histidine kinase and a response regulator. The bacterium E. coli uses a two component system called the phosphate signaling pathway to sense and respond to varying levels of phosphate in the environment.
The phosphate signaling pathway helps maintain the level of phosphate within the cell. Phosphate is important because it is a major component of DNA, RNA and cell membranes. In this system, a transmembrane histidine kinase, PhoR, is phosphorylated by ATP. PhoR donates this phosphate to a response regulator, PhoB, which autophosphorylates and activates transcription of the Pho regulon. The gene products of the Pho regulon help bring phosphate into the cell.
PhoB, the response regulator of the phosphate signaling pathway is a 26.3 kDa, 229 residue protein. To activate transcription PhoB must, in order, bind a divalent cation (Mg2+ ), autophosphorylate and form a dimer. PhoB can then activate transcription of the Pho regulon by binding DNA and interacting with RNA polymerase.
The goal of this project is to map the structure-function relationships of PhoB. We want to determine which amino acids act in each step of transcriptional activation. This knowledge will help to determine the molecular mechanisms of PhoB in the phosphate signaling pathway.
In this project mutations were introduced into the PhoB gene. These full length, loss of function proteins were sequenced for point mutations, over-expressed, purified and biochemically analyzed. Metal binding was measured using fluorescence quenching techniques and phosphorylation was measured using radio-labeled phosphorylation assays.
Metal Binding:
Tryptophan is an aromatic amino acid which normally fluoresces. The 54th residue of PhoB is a tryptophan. Trp 54 is near to the active site and is sensitive to what occurs there. When metal is bound, or phosphorylation occurs, the amount of fluorescence from Trp 54 decreases. By measuring this quenching, we can determine what is occurring at the active site.
L56S PhoB contains a mutation near the active site. Metal binding was measured using fluorescence quenching techniques. The KD of the L56S mutant is 0.91±0.19 mM for Mg2+, very similar to the 1.5±0.2 mM KD of wild-type PhoB showing that the L56S mutant bound Mg2+ similarly to the wildtype PhoB.
A second mutant, D53N PhoB, was analyzed using the same fluorescence quenching techniques. This mutation changed Asp 53, which normally binds metal in wild-type PhoB. No binding and no fluorescence quenching were expected for this mutant. Upon addition of Mg2+, D53N PhoB produced a change in fluorescence of less than 1%. This change may have been entirely due to background noise, which indicates the mutant, as expected, did not bind metal.
Phosphorylation:
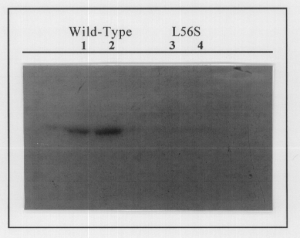
A preliminary phosphorylation assay was used to determine if L56S PhoB could auto-phosphorylate. Wild-type and L56S PhoB were incubated with 32P acetylphosphate (a phosphodonor). Wild-type PhoB displayed a great deal of auto-phosphorylation while L56S showed little or none (Fig. 1). Further phosphorylation assays will be performed to confirm these results. These preliminary results show that changes in the 56th amino acid allows metal binding to occur as in wild-type PhoB, yet prevents auto-phosphorylation. These results also confirm that the 53rd amino acid is necessary for metal binding. Using these procedures we will be able to characterize many mutants to discover how each amino acid affects the function of PhoB. By doing these types of experiments we can discover the molecular mechanisms of PhoB.
References
- McCleary, W.R. (1996) The activation of PhoB by acetylphosphate. Molecular Microbiology, 20,1155-1163. 163