Nathan La Monica and Dr. Dixon Woodbury, Physiology & Developmental Biology
SNAP-25, a protein that is found abundantly in the brain, is a key player in the process of releasing neurotransmitters. Vesicles, or small spheres of lipid membranes, contain neurotransmitters and have numerous proteins that extend outward from the vesicle. To release the neurotransmitters proteins on the vesicle bind with proteins on the pre-synaptic membrane causing the fusion of the two membranes. SNAP-25 along with Syntaxin (membrane bound protein) and Synaptobrevin (vesicle bound protein) work together to form a complex called the SNARE complex (Bowen, 2004). The current theory on how this complex works is that SNAP-25 has to be bound to the pre-synaptic membrane to participate in the SNARE complex. However, after finding that SNAP-25 naturally exists on the vesicle, Dr. Dixon Woodbury began studying to see if SNAP-25 can also act as a vesicle bound protein. Some of Dr. Woodbury’s preliminary data suggests that SNAP-25 may act in such a manner, an ORCA grant was awarded to continue the study of SNAP-25 acting as a vesicle bound protein.
A brief description of the methodology that I am using is necessary to understand the results. I create a lipid bilayer over a small hole which acts like the pre-synaptic membrane. By running a current through the membrane and adding modified synaptic vesicles (MSV’s), which are rat synaptic vesicles with a substance added, to the surrounding liquid I can tell when the vesicles fuse to the lipid bilayer. When an MSV fuses to the bilayer a voltage spike occurs on the computer measuring the assay.
Although the assay is straight forward once understood, it may still cause frustration. The ingredients that go into making the MSV’s are sometimes fickle. When one ingredient gets contaminated, oxidized, or too concentrated none of the vesicles work correctly. A couple times this past year the assay would be working great, and then one of our ingredients would spoil and the assay would stop working. When this happens basically the errant ingredient is determined by time consuming trial and error. We replace what we think is wrong, test the vesicles and if that does not work we replace the next ingredient.
The first step in the assay is to obtain synaptic vesicles, which we are able to get from extracted rat brains. Through several hours of processing and purifying we extracted several hundred microliters of purified synaptic vesicles suspended in a sucrose solution. Following the extraction of the synaptic vesicles we determined that to validity our results we would have three test batches. One batch would be boiled to denature all proteins and hopefully make the vesicles highly unlikely to fuse. Another batch would be treated with BoNT, which clips SNAP-25 from the vesicle. The final batch we would mock treat, thus acting like we treated it with BoNT, but just adding a simple saline solution to the batch leaving SNAP-25 intact. After we treated the synaptic vesicles we combined them with artificial vesicles, making modified synaptic vesicles.
Before we run the assay, the protein Syntaxin is added into the bilayer membrane. With this protein in the bilayer membrane all the proteins are present that are needed to make the fore-mentioned SNARE complex, but instead of SNAP-25 also being in the membrane it is on the mock treated vesicles. The boiled and treated SV’s act as a control. The results of experiments that were run are graphed in Chart 1.
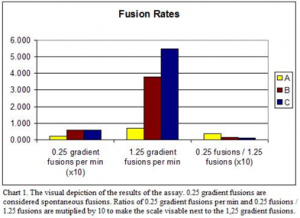
Batches A, B, and C are the boiled, treated, and mock treated vesicles respectively. The boiled MSV’s had far lower fusion rates than both the treated and mock-treated MSV’s. The fusions that occur during the 0.25 gradient are considered spontaneous fusions. We add a salt gradient to the solution to give a little energy to help the vesicles fuse. The 1.25 gradient adds a significant amount of energy and assists even more vesicles to fuse. The results of the set of experiments is not very convincing. The fusion rates ratio of the treated and mock treated vesicles are similar and do not provide a strong case for SNAP-25 acting as a vesicle bound membrane. The key number that is low is the mock treated 0.25 gradient fusions. These spontaneous fusions are actively involved with the Syntaxin that has been inserted into the bilayer membrane. The results that I obtained go against Dr. Woodbury’s preliminary results. A source of possible error is the incorporating of the Syntaxin, thus making the 0.25 gradient almost worthless. Another possibility is that the Syntaxin I used was either oxidized or too old. I used a batch of Syntaxin that was made back in 2007, which is satisfactory if the batch stayed frozen the entire time, but if it was thawed and refrozen multiple times then the Syntaxin may need replacement.
The next step in testing our hypothesis is to collect additional data so that we may have sufficient data to either prove or disprove our hypothesis. Our lab is in the process of securing more BoNT so that we may make up another batch of MSV’s. I will be using a newer batch of Syntaxin to see if the age of the Syntaxin had any negative effects on the experiments.
Overall, I learned a great deal about the process of working through problems. The data I collected is a portion of what is needed to publish a paper regarding our hypothesis. I am also preparing to present my results in a poster at the 53rd annual meeting of the Biophysical Society in Boston, MA. Now that I am familiar with the problems that I will face using this assay I feel confident that I will be able to quickly gather data so that our lab may continue at the forefront of the study of SNAP-25.
References
- Bowen, M.E., Weninger, K., Brunger, A.T., and Chu,S. “Single Molecule Observation of Liposome-Bilayer Fusion Thermally Induced by Soluable N-Ethyl Maleimide Sensitive-Factor Attachment Protein Receptors (SNAREs).” Biophysical Journal. 87 (2004): 3569-3584.
