David Holt and Dr. Robert Seegmiller, Physiology and Developmental Biology
Introduction
Spondyloepiphyseal dysplasia congenita (SEDC) is a recessive human genetic disorder characterized by skeletal abnormalities and early onset osteoarthritis (OA).1 Type II and the alpha 3 chain of type XI collagen together are integral in the development of normal articular cartilage (AC) and are both encoded by the Col2a1 gene.2 Spondyloepiphyseal dysplasia congenita (sedc) mice have a missense mutation of the Col2a1 gene within the triple helical region of type II collagen, affecting its production, leading to development of abnormal AC.1 We have identified degenerative indicators in sedc mice such as fissures in the AC cartilage and a lack of extracellular matrix (ECM). These problems are related to the sedc mutation which observationally causes collagen producing cells (chondrocytes) to form a large space around the cell (pericellular space) due to an apparent inability to modify structural proteins needed to construct the ECM. Two key protein groups in the formation of AC are type II collagen and proteoglycan. Type II collagen is responsible for the development of fibers that structurally support the ECM. Proteoglycans are a group of proteins that help to make the cartilaginous tissue resilient by acting as a backbone for the bonding of molecules like keratin sulfate and chondroitin sulfate that make the tissue stiff by retaining water. The ECM is the structural foundation of normal cartilage and is significantly diminished in sedc mutant mice when compared to normal wild-type controls. The lack of ECM may be associated with deceased amounts of type II collagen and other significant proteins in the ECM. MMP-13 is one of the key enzymes involved in the degradation of type II collagen and can be seen using immunohistochemical (IHC) techniques to determine if it is involved in ECM destruction.3,4 This study has a two-fold purpose: first, to characterize the ECM of AC in sedc mice by identifying and localizing the major structural proteins present, and second, to determine the mechanism involved in the lack of ECM and the degeneration of AC. The molecular implications of the sedc mutation have yet to be studied and are anticipated to provide new understanding relating to mechanisms that cause OA. As a result, data collected from this study may have significant clinical implications because it could help advance the search for a more effective treatment of OA through the development of therapeutic drugs.
Methods
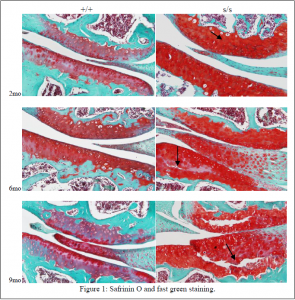
This study was done using two types of sedc tissue samples, one from wild-type (+/+) and the other from homozygous (s/s) mice for a total of six samples from each of the age groups mentioned below. Genotypes were obtained using PCR analysis for all of the mice used in this study. The study was performed using age matched +/+ controls at two, six, and nine months of age to show progression of AC degeneration. Antibodies were used against type II collagen and MMP-13 to perform IHC analysis of tissue sections to detect the presence and location of the structural protein and destructive enzyme. Sections were also stained with Safrinin O and Fast Green to show the presence and localization of proteoglycan. After all staining was completed the samples were photographed for comparative analysis using a light microscope.
Results
Safranin O staining showed an apparent increase in the production of proteoglycan in the AC of the s/s mutant knee joint when compared to the wild type control at all ages. This was acompanied by obvious signs of fissuring and AC degeneration at six and nine months (See Figure 1). IHC staining showed an increased amount of type II collagen in the pericellular space of the chondrocytes in the s/s mice compaired to the wild-type. This was shown at two, six and nine months of age and is an indication of the breakdown of the ECM. IHC also showed an increased production of MMP-13 in the s/s mutant mouse at six and nine months of age.
Conclusions
From histological analysis it is apparent that the sedc homozygous mutant displays significant histological differences in comparison to the wild-type control. A significant indicator of early onset OA is the increased amount of Safranin O positive staining in the pericellular spaces and the overall AC of the s/s mouse compared to WT. The upregulation of proteoglycans may be a coping mechanism to compensate for the breakdown of the ECM. This was also accompanied by significant fissuring and clustering of cells in the s/s. IHC staining showed an abnormal amount of type II collagen staining in the pericellular space of the s/s mouse. Type II collagen is primarily found in the ECM and is not ususally present in the pericellular space, therefore, this is an indication that there is a breakdown of the ECM. The staining of type II collagen in the pericellular space and its amorphous appearance suggests normal polymerization of type II collagen is not occurring or it may suggest an abnormal breakdown of collagen fibrils. These data suggest that sedc mutant mice are incapable of correctly regulating proteins integral in the formation of ECM which may be due to the degradation of type II collagen. This defective ECM may be caused by the up-regulation of destructive enzymes which therefore lead to the premature degradation of AC. Thus, the sedc mouse presents itself as a viable murine model for the study of OA.
The data presented here is only preliminary; more data must be gathered before this project is ready to be submitted for publication. We encountered problems with our breeding and have not gathered enough animals to have significant sample sizes and must wait for additional animals to mature. We also plan to do IHC on other proteins we believe play a significant role in AC degeneration.
References
- Donahue LR, Chang B, Mohan S, Miyakoshi N, Wergedal JE, Baylink DJ, Hawes NL, Rosen CJ, Ward-Bailey P, Zheng QY, Bronson RT, Johnson KR, Davisson MT. “Missense Mutation in the Mouse Col2a1 Gene Causes Spondyloepiphyseal Dysplasia Congenita, Hearing Loss, and Retinoschisis”. J Bone Miner Res. 18.9 (Sep 2003), 1612-1621.
- R.R. Rodriguez, R.E. Seegmiller, M.R. Stark and L.C. Bridgewater. “A type XI Collagen Mutation Leads to Increased Degradation of Type II Collagen in Articular Cartilage”. Osteoarthritis and Cartilage 12 (2004), 314–320.
- L. Xu, C.M. Flahiff, B.A. Waldman, D. Wu, B.R. Olsen and L.A. Setton et al., “Osteoarthritis-like Changes and Decreased Mechanical Function of Articular Cartilage in the Joints of Mice with the Chondrodysplasia Gene (cho)”. Arthritis Rheum 48 (2003), 2509–2518.
- T. Sadowski and J. Steinmeyer. “Effects of Non-steroidal Anti-inflammatory Drugs and Dexamethasone on the Activity and Expression of Matrix Metalloproteinase-1, Matrix Metalloproteinase-3 and Tissue Inhibitor of Metalloproteinases-1 by Bovine Articular Chondrocytes.” Osteoarthritis and Cartilage. 9 (2001), 407-415
- B.D. Bomsta, L.C. Bridgewater, and R.E. Seegmiller. “Premature Osteoarthritis in the Disproportionate Micromelia(Dmm) Mouse”. Osteoarthritis and Cartilage 14 (2006), 477-485.
