Devin Holden and Dr. Laura Bridgewater, Microbiology and Molecular Biology
Bone morphogenic protein 2 (BMP2) is a protein that is secreted outside many different types of mammalian cells and has been implicated in the pathogenesis of various forms of cancer. Recently, a variant of this protein that is localized to the nucleus (nBMP2) of the cell was discovered and characterized “i”. In order to further characterize this novel variant of BMP2 a microarray analysis was performed to assess what influence nBMP2 has on the expression levels of different genes in a typical mammalian cell. This analysis revealed a list of several genes whose expression levels are potentially altered by nBMP2. From this list I chose to focus on five genes, all of which have previously been implicated in cancer. I performed experiments to confirm the findings of the microarray analysis, and am working towards testing whether or not nBMP2 is binding to the DNA in order to exert its regulatory effects.
Microarray analysis of gene expression in cells is considered a preliminary analysis. Results obtained from microarray analysis must be confirmed by another experimental procedure in order to be considered conclusive. The aim of the first part of my project was to confirm the microarray results for the five genes that I had chosen. This proved to be more of a challenge then I had originally anticipated. The first step in the experimental procedure to confirm the microarray analysis was to transfect cells derived from mice with one of the five genes that I had chosen. This proved to be the most challenging part of my project. Normally, transfecting mammalian cells is not a difficult experimental procedure to perform successfully, but in my case the first few months were spent working with minimal success. My mentor and I tried many different approaches in order to troubleshoot the problem and after a few months I began seeing positive results, although the procedure was not working well enough to continue to the next step. I optimized the experimental protocol in order to obtain results of sufficient quality to proceed.
Once results of sufficient quality were obtained I was able to move onto the next step, which involved the use of an experimental procedure known as quantitative reverse transcription polymerase chain reaction (quantitative RT-PCR). In order for this experimental procedure to produce quality results it also needed to be optimized. I performed the needed optimization and obtained some preliminary data. The data revealed that one of the five gene’s, Sphingosine-1-phosphate lyase 1 (SPL-1), expression level was indeed altered by nBMP2. At this time my mentor approached me and explained that she had a manuscript that had been submitted to be published, but which had been sent back because it needed some revisions. The manuscript also needed some additional experimental procedures to be performed in order to ensure that the findings of the manuscript were conclusive. She explained that the experimental procedures which needed to be performed were identical to the ones I was currently using for my ORCA project, but that I would transfect a different cell line using a different gene, called Lc-Maf. She asked if I would be willing to perform the experiments with the goal of resubmitting the manuscript for publication with the results from my experiments, and I accepted.
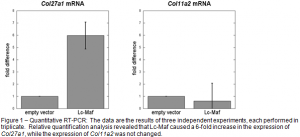
Lc-Maf is a protein that regulates the expression of many different genes in mammalian cells. The goal of the experiments I performed was to test whether or not Lc-Maf altered the expression of two genes, Col27a1 and Col11a2, which are involved in orchestrating the changes involved in cartilage cells transitioning into bone cells during skeletal development. In order to accomplish this I transfected mammalian cells derived from rats with the Lc-Maf gene. Because I was using a different cell line than I had used previously, the experimental procedure had to be optimized again. This was a time consuming procedure, but I was able to obtain some positive results. Once positive results were obtained I moved to the next step which again involved quantitative RT-PCR. I performed the experimental procedure and was able to obtain quality data (Fig. 1). The data revealed that Lc-Maf increases the expression the Col27a1 gene, but does not significantly affect the expression of Coll11a2 gene. These results were incorporated in the manuscript, and we are currently in the process of choosing an appropriate journal to submit the manuscript to. I have also submitted an abstract using the data relating to the experiments with the Lc-Maf gene to the American Society for Biochemistry and Molecular Biology with the goal of presenting a poster at their annual meeting in April of 2009.
This research project has been a very valuable learning experience and will no doubt benefit me greatly in my future pursuits. My goal is to make as much progress as possible on this research project during my last semester. Currently, I am continuing work on the original five genes that I chose and how their expression levels are affected by nBMP2. We were forced to purchase new mouse cells to transfect with nBMP2 because the original cells that I successfully transfected would no longer transfect for unknown reasons. I am in the process of optimizing the transfection protocol for these new cells. If I am able to successfully transfect these cells I will then perform quantitative RT-PCR. If that step is successful, I will then move onto the next step and begin assessing whether or not nBMP2 binds directly to the DNA to exert its regulatory effects on the five genes I have chosen.
References
- Mayo J. “Characterisation of a novel nuclear variant of Bmp2 and coordinate regulation of Col11a2 and COL27A1 by the transcription factor Lc-Maf” Dissertation (2007) Provo, UT: Brigham Young University
