Liz Gibbons and Dr. John Bell, Physiology and Developmental Biology
The purpose of my project was to further study cell membrane changes during apoptosis, or programmed cell death. This was an extension of the research being done by Dr. Bell and my fellow students on the susceptibility of cells to the enzyme secretory phospholipase A2 (sPLA2). This enzyme hydrolyzes cell membranes, but only those with certain lipid properties. In general, only cells that are dead, injured, or dying display significant susceptibility to sPLA2 while healthy cells resist hydrolysis (Nielson, 2000). We use fluorescent probes to study these membrane lipid properties and how they change as the cells undergo apoptosis. These membrane changes can have important pathological implications. In cancer, for example, cells which should have entered programmed cell death and been made susceptible to hydrolysis by sPLA2 have somehow become resistant. If we better understand the properties of membranes which induce susceptibility, we may be able to understand the resistance of cancer cells and find ways to treat it. Likewise, our research seeks to better understand some of the implications, including potential inflammatory responses, of chemotherapy designed to elicit apoptosis.
My study specifically focused on a fluorescent probe called merocyanine 540 (MC540). MC540 is a lipophilic probe whose emission increases in intensity and shifts in wavelength when it binds to cell membranes. Emission intensity decreases as MC540 is excluded from the membrane (Langerner and Hui, 1993). Increased MC540 fluorescence has been observed when cells become susceptible to sPLA2 (Bailey 2007). We have interpreted this elevation of fluorescence to indicate an increase in the spacing among adjacent phospholipids in the membrane, which might allow them easier access to the enzyme. Thus far, we have not been able to identify the cellular mechanism leading to this increased MC540 fluorescence during apoptosis. One likely possibility is that the exposure of phosphatidylserine (PS) is responsible for the membrane changes that MC540 is measuring. PS is a negatively-charged phospholipid that is localized on the inner leaflet of the phospholipid bilayer in healthy cells. During apoptosis, PS “flip-flops” to the outer leaflet, exposing this negative charge. It seems logical that these negative charges would repel each other and thus create greater spacing among the lipids. To test the hypothesis that this increase in MC540 fluorescence is strictly due to the flip-flop of PS, I used a drug called dibutyryl cyclic AMP (dbcAMP) which has been reported to signal the apoptotic pathway with less PS exposure than that observed with other apoptotic inducers (Neilson 2000).
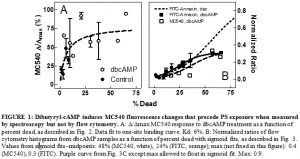
I began by measuring MC540 fluorescence in dbcAMP-treated S49 mouse lymphoma cells using fluorescence spectroscopy. These experiments repeatedly showed increased fluorescence intensity, and thus greater lipid spacing, compared to controls. I plotted this increase in MC540 fluorescence against the percentage of the cell population that was dead to represent how far into the apoptotic process the cells had advanced (see Figure 1, panel A). I fit the data with a hyperbola whose Kd was 6% dead, indicating that this increase in MC540 fluorescence was an early event in apoptosis induced by dbcAMP. This seemed to contradict my hypothesis that the MC540 fluorescence was due to PS flip-flop since this drug had been reported to exhibit low levels of PS exposure.
To confirm this result, I needed to show that PS exposure could not be responsible for the increased spacing in my samples. Therefore, I used flow cytometry to measure the fluorescence of two probes concurrently: MC540 to measure lipid spacing and FITC-labeled annexin-V to measure PS flip-flop. The results (see Figure 1, panel B) seemed to contradict the results I saw in the fluorescence spectroscopy experiments. Although the samples did show reduced PS exposure when compared to other apoptotic inducers (such as dexamethasone, also shown in panel B) as reported, we did measure PS flip-flop that preceded the increase in MC540 fluorescence. These data (percentage of the cell population exhibiting either MC540 or FITC-annexin fluorescence above the background level) were also plotted against percent of population dead and fit with sigmoid curves. The midpoint of MC540 and FITC-annexin were 48% and 24% dead respectively. This was an interesting result, since the Kd of MC540 fluorescence was 6% as measured by fluorescence spectroscopy. These results suggest that MC540 spectral changes are independent of and precede PS exposure, but that MC540 changes evident from flow cytometry follow PS exposure. Although this is a step in the right direction, more experiments will be needed to further determine the role of PS flip-flop in MC540 fluorescence changes.
References
- Bailey RW, et al. (2007) Biophys J. 93, 2350-62.
- Langner, M., and Hui, S. W. (1993) Biochi. Biophy. Acta. 1149, 175-179
- Nielson, K. H., Olsen, C. A., Allred, D. V., O’Neill, K. L., Burton, G.F., and Bell, J. D. (2000) Biochim. Biophys. Acta. 1484, 163-174
