Nate Keplinger and Dr. Byron Adams, Biology
Abstract
Slugs and snails are pests that destroy various food crops throughout the entire world. To control their outbreak, Europeans have utilized the nematode Phasmarhabditis hermaphrodita, a mollusc pathogen. This nematode has not been introduced into US agriculture because it has not been found in a mollusc pathogenic stage in the United States and could have detrimental non-target effects. Thus, slug and snail control methods in the US are currently limited to petrochemical methods of pest management. I have recently discovered an unidentified species of the genus Phasmarhabditis. that rapidly and consistently kills slugs, snails and grubs. I propose a more careful study of their biocontrol potential by testing their virulence on different snail species through titration (dosage) tests as well as their mutualistic association with bacteria. These tests will establish the infection density required to kill slugs, an important step towards developing the nematodes for pest control applications in the US as an alternative to traditional, chemical-based pesticides.
Introduction
Slugs and snails are serious agricultural pests throughout the US. The discovery of Phasmarhabditis in the United States could help reduce the cost of pesticides to mollusc plant pests as well as reduce harmful chemicals from entering food and water systems. These nematodes could be introduced into the United States agricultural system as well as other agricultural systems throughout the world.
The nematode Phasmarhabditis was first discovered in 1859. The specific species Phasmarhabditis hermaphrodita has been used to infect various species of molluscs with many different results. Some mollusc species are not affected by the presence of the nematode while others are only susceptible to nematode parasitism if the mollusc is in its juvenile stage or if the mollusc is fairly small or too large. There is also a large time window of 4-21 days that determines the mortality rate of the mollusc. Lastly, the Phasmarhabditis spp. studied thus far are very temperature specific. They can effectively kill molluscs anywhere from 5°C-17°C, but die at temperatures exceeding 35°C.
I have already shown, through various experiments, that the Phasmarhabditis sp. I discovered in Utah successfully kills slugs, snails, and grubs. In order to test how effective they will be as a molluscide, I am going to performd different dosage response assays of nematodes to different species of molluscs. I hypothesize that this species of Phasmarhabditis nematodes will effectively kill the molluscs within the 4-21 day window utilizing different dosages. These tests will help to prepare the Utah Phasmarhabditis sp. nematode for the United States agricultural system.
Methods
Nematodes were first isolated by sampling random soil around BYU’s campus. The soil was then passed through a sugar centrifugation process using a 15 inch U.S. Standard Testing Sieve with a 38µm screen. After extracting the nematodes from the solution, they were used in baiting Galleria mellonella grubs sitting on top of 5.5 cm filter paper inside of a 55 mm Petri dish. The grubs were stored at 20° C until mortality. Once dead, the grubs were placed into a White trap system consisting of a 90 mm Petri dish with a smaller 40 mm Petri dish inside. The smaller Petri dish was then covered with 7.5 cm filter paper and filled up with water until well moistened.
Once the nematodes emerged, they were pipetted out into 40 ml culture flasks with only 10 ml of water so they could be stored properly and not leak. The nematodes were stored at 15°C. Random nematodes were selected from the flasks to be surface sterilized and crushed to extract inner bacteria. The bacteria were plated onto a 90 mm Petri dish agar plate using a 3 mm sterile loop. The Petri dish was then stored at 37°C to culture the bacteria. The bacteria were analyzed using standard gram staining materials and microscopes.
Snails and slugs were collected from various local gardens in Provo, Utah. The two species utilized in the experiment were Helix aspersa (snail) and Limax maximus (slug). These were chosen for various reasons:
1. They were easy to find.
2. They both are an abundant species in Provo, Utah.
3. As can be noted in the table above, scientists have only been able to kill juveniles of the specific snail species and had no success killing the specific slug species.
The molluscs were stored inside of a styrofoam container (18 in. x 30 in. x 15 in.) for 10-15 days and fed on local shrubbery as well as cabbage and baby carrots. The specimens were stored for this time period to avoid any prior bacterial or chemical infection so the nematodes could kill the molluscs without any aid. In preparation for infection, 5 ml sample containers were coated with FLUON® (Whitford Dispersion, UK) to create a non-stick surface so the molluscs would remain inside of the infective area. A 5.5 cm piece of filter paper was then placed on the bottoms of the flask. Each mollusc specimen was weighed and placed into the container with concentrations of 5000, 2000, 1000, 500, and 200 nematodes per 5 ml dH2O (average of 50 nematodes per 5 mm diameter (20 μl) of nematode infested dH2O). The molluscs were stored for up to 21 days at 15-19°C with a 14:10 l/d period and a humidity level of 34%. The molluscs were not fed for the first 24 hours so the infection period would not be hindered.
After two days, the molluscs were fed a 1 in. diameter piece of cabbage which was changed every 2 days consecutively. The molluscs were analyzed on a daily basis to look for any deterioration of their body, any swelling, discoloration, or mortality. Dead specimens were removed and allowed to dry for 2 days before being moved into a White trap system using the 5 ml container, now sanitized with bleach water, a 40 mm petri dish, 5.5 cm filter paper, and enough dH2O to maintain a moist environment. White trapped molluscs were then analyzed on a daily basis to see if any nematodes emerged from their corpses. If nematodes were present, they were washed three times by pouring them through a funnel into a 13 ml falcon tube, filling the tube up with 10 ml of dH2O, and centrifuging at 3000 rpm’s for 3 minutes. The washed nematodes were then pipetted out using a 100μl-1000μl into a 40 ml culture flask with 10μl of dH2O and stored at 15°C so they could be utilized for further titration and bacterial studies. Molluscs that survived past day 21 were removed from the experiment. Controls were used by placing the molluscs into an identical system but with 5 ml of dH2O without nematodes.
Results
Phasmarhabditis sp. Nematodes were luckily and successfully extracted from 1 of the 10 soil samples from around campus. After infecting the Galleria mellonella with the Phasmarhabditis sp., the grubs’ average mortality rate was 7 days. It took another 7 days for the nematodes to emerge from the grubs after placing them in the White trap system. The DNA analysis showed the 28s ribosomal RNA region base pairs to be around 700-800 bp. These basis were Blasted on the NCBI website and found to be 100% related to a Phasmarhabditis sp. from New York (EM434). The nematodes have been successfully cycled through this system for 1.5 years.
After randomly sampling the nematodes, surface sterilizing them (placing them into a diluted bleach solution for 20 seconds) and crushing them the bacteria from inside of their gut was successfully cultured on agar plates. The bacteria was then gram stained and examined under a microscope. The analysis of the bacteria resulted in a gram negative bacteria that is coccal and rod in shape as well as having the appearance of being pleomorphic (not separating after fusion replication stage so it appears that there are two bacterium together). The DNA analysis did not come back successful after performing PCR on the 16s ribosomal RNA regions of the bacteria. However, the bacteria’s physical characteristics are identical to those described in the identification of a mutualistic bacteria of Phasmarhabditis sp. named Moraxella osloensis.
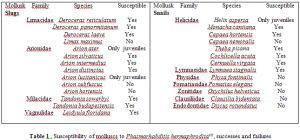
After dosage experiments were tested on Helix aspersa and Limax maximus, it can be concluded that the Phamarhabditis sp. is more successful at killing the Helix aspersa snail instead of the Limax maximu slug, which correlates with previous studies. However, the Utah Phasmarhabditis sp. kills both juveniles and adults of the snail species. As can be see in figure 1 the snails died at every titration rate of infection and were killed at almost the same percentage throughout the entire experiment. The standard deviation shows that the average mortality % per dosage assay was around 85% and that the all of the mortality % per dosage assay are statistically significant.
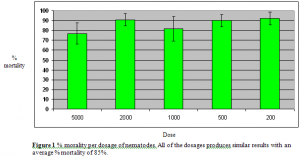
After understanding that all of the dosage response assays had a % mortality average f 85%, it was important to deduce if there was a correlation between the weight of the snail specimens and the dosage. The snails that were part of the experiment weighed between 0.65 (g) – 11.36 (g). Mortality appeared at all weight levels. According to figure 2 on the next page, the R-value of the scatter plots are so small that there is no statistical correlation between the weight of the specimens killed and the dosage of nematodes. Only three graphs (dosages of 5000, 1000, and 200 nematodes) are pictured because they are the high, middle, and low dosage response assay tests performed. The other dosages of 2000 and 500 demonstrated the exact conclusion of no correlation between weight of the specimens killed and the dosage of nematodes.
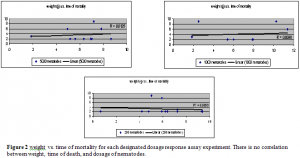
After establishing credible and statistical evidence that there is no weight:dosage correlation, it is was important to note the number of days that it took for the snail specimens to die for each of the four experiments performed.. According to the data shown in figure 3, the snails died anywhere between 2-9 days, with most of the specimens dying on day 2. The samples were control corrected. The error bars from statistical analysis appear a little skewed because some of the experiments performed had all of their specimens die on one day and none on the other. However, the data is still statistically significant
Conclusion and Future Directions
After studying the Phasmarhabditis sp. nematode from Utah, it is possible that the nematode could be used as a safe, efficient, and effective molluscicide. It appears to kill molluscs at a faster rate than the European species and also appears to kill a wider range of specimens ranging from juveniles to adults. Further studies will have to be performed on the Helix aspersa snail and the Limax maximus slug, as well as other mollusc specimens, to determine the nematode’s virulence specificity. More careful controls will be performed during these experiments as well as smaller dosages of nematodes since dosages between 5000 and 200 nematodes per 5 ml of dH2O killed at a relatively similar percentage. Once more data has been collected, it is critical to test the nematode’s virulence in field experiments by beginning with soil baiting and moving out to local gardens and fields. These experiments will have to be repeated several times to establish further credibility on the Phasmarhabditis sp. nematode or it to be marketed as a successful molluscicide.
Not only will it be important to further understand the nematode’s virulence, but it will also be important to identify and characterize the mutualistic bacteria to assure safety in food and water intake. Once the bacteria is properly and completely identified, the nematodes will be cultured in the bacteria (fermentation culturing) to establish an easier method of culturing for mass production.
References
- Barker GM, Molluscs as Crop Pests. CABI Publishing, Wallingford,UK (2002).
- Robbie R,Cyrille V, Parwinder SG, Jamie FR, and Michael JW, Biological Control of Terrestrial Mollusks Using Phasmarhabditis hermaphrodita –progress and prospects. Pest Management Sci 63:1153-1164 (2007).
- Schneider A, U¨ ber eine Nematodenlarvae und gewisse Verschiedenheiten in den Geschlechtsorganen der Nematoden.Zeitschrift f¨ur Wissenschaft Zoologie 10:176–178 (1859).
- (Ibid.)
- Wilson MJ, Glen DM, and George SK, The rhabditid nematodePhasmarhabditis hermaphrodita as a potential biological control agent for slugs, Biocont Sci Technol, 63:503–511(1993).
- Robbie R,Cyrille V, Parwinder SG, Jamie FR, and Michael JW, Biological Control of Terrestrial Mollusks Using Phasmarhabditis hermaphrodita –progress and prospects. Pest Management Sci 63:1153-1164
- Robbie R, Cyrille V, Parwinder SG, Jamie FR, and Michael JW, Biological Control of Terrestrial Mollusks Using Phasmarhabditis hermaphrodita –progress and prospects. Pest Management Sci 63:1153-1164 (2007).
- Li Tan, M.S., Virulence mechanisms of the nematode Phasmarhabditis hermaphrodita and its associated bacteria Moraxella osloensis to the gray garden slug Deroceras reticulatum. Dissertation, Ohio State University (2002).
