Matt Gruwell and Dr. Michael Whiting, Zoology
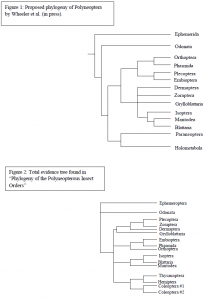
Despite recent morphological and molecular investigations, phylogenetic relationships among the Polyneoptera remain nebulous. If this group is monophyletic, then it represents one of the largest and morphologically most diverse of all insect radiations. The Polyneoptera includes the orders Orthoptera, Phasmida, Plecoptera, Embioptera, Dermaptera, Grylloblattaria, Isoptera, Blattaria, and Mantodea, and may include the Zoraptera. 18S ribosomal DNA and Cytochrome Oxidase II (CO II) protein coding DNA sequence data from multiple taxa representing the Polyneoptera, as well as multiple outgroup taxa, were combined with the morphological data to assess the phylogeny of this group. The results of a recent analysis (fig. 1) by Wheeler et al. (in press) are contrasted with these results.
A morphological matrix of all relevant characters was adapted from Wheeler et al. (in press) and put into Winclade. This matrix was spawned into Nona (Goloboff 1994) to determine phylogenetic relationships with respect to morphology.
DNA sequences were both generated and taken from Genbank, a website in which researchers make available DNA sequences from projects they have already completed. Specific regions of DNA were amplified by making use of phenyl/chloroform extraction and PCR (polymerase chain reaction) techniques. Once the DNA was amplified and cleaned, samples were cyclesequenced and dyed with Big-Dye. During cycle-sequencing the piece of previously amplified DNA is amplified again, but in this process the amplification is randomly stopped by a dye dichlororhodamine terminator which attaches a fluorescent dye to a the last dideoxynucleotide via a linker. The fluorescent labeled base pair is read as each piece passes size wise through a laser via electrophoresis, in the 377 Automated DNA Sequencer.
Once data sets were compiled, sequences needed to be aligned for phylogenetic analysis. The COII gene alignments were done in Sequencer and proofed by eye. This method was chosen because COII is a protein-coding gene and therefore alignment must be sensitive to the codon positions, any alignment that inserted gaps within the three base pair codon would yield incorrect alignments. 18s data was aligned in MALIGN (Wheeler 1994) by inserting five conserved regions the program. Once aligned the five sections were then joined and analyzed with maximum parsimony in PAUP (Swofford 1993). All data sets were analyzed individually and combined to generate a total evidence topology.
The total evidence tree (fig. 2) provided the most information toward understanding the evolution of the Polyneopterous insect orders. Within that clade the group Polyneoptera did not demonstrate monophyly with respect to the outgroup members from Paraneoptera and Holometabola. Instead the clade of Polyneopterous and non-basal outgroup taxa was unresolved, leaving the origin of Polyneoptera unknown. Within this clade of the Polyneoptera and outgroup taxa, the orders thought to be within Polyneoptera always appear in clades with other Polyneopterous orders and are not found in the clade of outgroup taxa. This weakly supports the possible monophyly of Polyneoptera.
Relationships of orders within Polyneoptera show some discrepancies with that of the Polyneoptera clade presented by Wheeler et al. (in press). The first contradiction is that Wheeler demonstrated a clade of Orthoptera, Phasmida, Plecoptera and Embioptera yet this total evidence tree suggested that Plecoptera was a sister group to Zoraptera and in a clade with Dermaptera, and Grylloblatteria. Another difference is shown where Phasmida and Embioptera are represented as sister taxa rooted to Orthoptera instead of Phasmida and Orthoptera being sister taxa. This new evidence is in support of Maxwell (in press) who demonstrated Embioptera to be the sister group to Phasmida. One similarity is shown in the group Dictyoptera (Mantodea, Blattaria, and Isoptera). However, Wheeler et al. (in press) show this group to be rooted within another clade with other orders and this total evidence tree suggests otherwise, but the lack of resolution makes it impossible to make a hypothesis as to the relation of Dictyoptera with other taxa.
This study is a first step taken towards the understanding of this very diverse group. To further determine the relationships within this group accurately, these, and possibly many other, steps need to be taken: 1) More taxa must be sampled, at least 10-20 specimens from every order representing each family within every order. 2) More basal and non-basal outgroups need to be included. These will force the unique characters of the Polyneoptera to demonstrate the monophyly of the group if it is monophyletic. 3) More data needs to be extracted from each specimen. For the project COII seemed moderately useful but due to its rapid evolutionary change it did not appear extremely useful in deciphering ordinal relationships. 28s, EF1-alpha and other genes could be explored to make the analyses more thorough. 4) More detailed morphological work would prove useful. In the matrix comprised by Wheeler et al. (in press) the morphological data was coded at the ordinal level. If characters were used to coincide with specific families used in the molecular analysis it would improve accuracy in combining data sets.