Rebecca Bradshaw and James Johnston, Health Science Department
Introduction
Asthma is the most common chronic disease among children worldwide, and the third-ranking cause of hospitalization for children under 15 years of age.(1-2) While the exact causes are unknown, studies show that having a genetic predisposition coupled with early-life allergen exposures increases the likelihood of developing asthma.(3) For inhalation exposure to occur, allergens must be airborne and must have an aerodynamic particle diameter of 10μm or less (PM10). Particles 10μm or less can penetrate to the bronchial region of the lungs. In sensitized individuals, inhaled allergens that reach the bronchi can initiate a hyper-responsive immune reaction, resulting in airway constriction, wheezing, and shortness of breath. House dust has been implicated as a major source of exposure to allergens. While low levels of allergens can be found floating in the air inside the home, we hypothesize that childhood allergen exposures occur primarily through the re-suspension of settled dust found in reservoirs such as carpets, bedding, and upholstered furniture. Furthermore, this dust re-suspension occurs in the immediate vicinity of the child while playing on the dust reservoir and is dependent on the type of play and level of activity. However, few studies have considered how children’s activity levels influence their PM10 dust exposures. Most studies measure allergen levels in settled house dust, and use this measure as a surrogate of exposure. Price et al. (1990), however, found that reservoir carpet sampling is not a good indicator of inhaled antigen load.(4) Personal breathing zone samples collected within 6 – 9 inches of the nose and mouth are preferred when conducting exposure assessments because contaminants are collected from air representative of that entrained into the lungs during inhalation; however, this method is expensive and difficult to use in large studies. Development of a mathematical exposure model will help identify how reservoir dust load, ambient PM10, and physical activity contribute to a child’s breathing zone PM10 exposure. We hypothesize that by adding ambient PM10, and the child’s activity level, we can improve the prediction of breathing zone allergen exposures. If the model is more predictive than reservoir sampling alone, future studies could refine their exposure estimates without having to collect personal breathing zone samples.
Methodology
Children ages 7 – 11 (N = 46) will be recruited through personal contacts and flyers at BYU. Participants’ breathing zone PM10 exposures will be monitored over a 2-hr period for each child while the child is in his/her home. Breathing zone PM10 will be measured by having the child wear a sampling vest fitted with a TSI SidePak AM510 (TSI, Inc., Shoreview, MN, USA) personal exposure monitor. The SidePak will pre-filter the air using a 10μm impact separator, and exposures will be recorded every 5 seconds during the sampling period. Reservoir dust samples will be collected using a Duststream® dust collector with a 40 μm filter (Indoor Biotechnologies, Charlottesville, VA). Reservoir dust will be collected from carpets, bedding, and upholstered furniture in the home by vacuuming 1m2 on each surface for 3 min. Reservoir dust will be sieved through a #50 wire mesh (300μm sieve). Background PM10 levels in the home will be collected by placing a TSI SidePak AM510 with a 10μm impact sampler in a stationary location in a main living area of the home (living room). Children’s physical activity will be recorded on a GoPro camera, and later coded according to pre-determined criteria. Physical activity level, reservoir dust load, and background PM10 will be used to develop an equation to more accurately predict breathing zone PM10 exposure (Equation 1). We will compare this new prediction equation to the current method which uses reservoir dust only (Equation 2).
Equation 1
Estimated PM10 Exposure = ß0 + ß1(reservoir dust load) + ß2(physical activity) + ß3(background PM10)
Equation 2
Estimated PM10 Exposure = ß0 + ß1(reservoir dust load)
Comparing these two equations will allow us to create a more accurate method for estimating children’s house dust exposure. Statistical analysis will include R-squared for the p-variable model, F statistic for comparing full and restricted models, mean squared error for the p-variable model, and Mallow’s C for the p-variable model. Analyses will be performed using SAS software, version 9.3 (SAS Institute Inc., Cary, NC, 2010).
Results
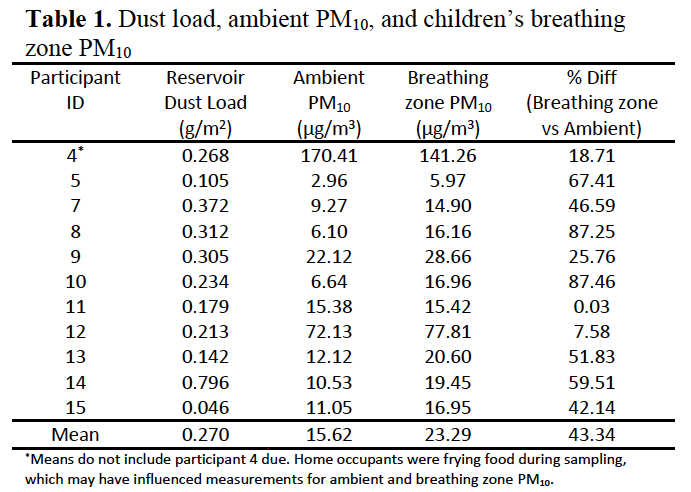
Data collection is still underway for this study, and the results reported here are preliminary. For all but one of the 11 participants sampled to date, breathing zone PM10 was higher than ambient PM10 (Table 1). PM10 concentrations were highest for participant 4 for both breathing zone and ambient measures. During sampling for this participant, home occupants were frying food which created visible smoke in the home. Due to this confounding factor, data from participant 4 was not included in calculations for mean ambient and breathing zone values. With participant 4 dropped, average breathing zone PM10 was 43.34% higher than ambient PM10. Reservoir dust load was calculated as the mean of the 4 dust samples collected in the home. Dust loads in homes ranged from 0.046 – 0.796 g/m2. From the data collected thus far, reservoir dust load is poorly correlated with ambient PM10 (r = .07) and breathing zone PM10 (r = .01). The Go-pro videos have not yet been coded.
Discussion/Conclusions
These preliminary results confirm previous studies suggesting that breathing zone PM10 exposures are often higher than ambient PM10 exposures. This is a good indication that the amounts of PM10 entrained into the lungs should not be estimated from ambient air samples alone, as this will likely under-estimate true exposure. While we have not coded the videos for physical activity level, our preliminary findings suggest that resuspension of house dust likely explains the significantly higher breathing zone concentrations found among the participants, and that physical activity is an essential factor to consider when predicting children’s house dust exposures.
References
- Hall, M.J., DeFrances, C., Williams, S., Golosinskiy, A., Schwartzman, A. (2007). National Hospital Discharge Survey. Retrieved from: http://www.cdc.gov/nchs/data/nhsr/nhsr029.pdf
- Asher I., Pearce, N. (2014). Global Burden of Asthma Among Children. The International Journal of Tuberculosis and Lung Disease, 18(11), 1269-78.
- National Heart, Lung, and Blood Institute, National Institutes of Health: National asthma education and prevention program. Expert panel report 3: Guidelines for the diagnosis and management of asthma. (2007) Retrieved June 1, 2012 from http://www.nhlbi.nih.gov/guidelines/asthma/asthgdln.pdf
- Price J., Pollock I., Little S., Longbottom J., Warner J.: Measurement of airborne mite antigen in homes of asthmatic children. The Lancet. 336:895 – 897. (1990).