Connor Woodward and Brent Nielsen, Microbiology and Molecular Biology
Introduction
Dna2 is a protein that is known to be essential for DNA replication in human and yeast mitochondria. In these organisms it acts as both a helicase and an endonuclease. In the model plant Arabidopsis thaliana an ortholog of Dna2 exists but has never been studied. Two questions were answered in this study. The first question is “What would happen to A. thaliana plants that lacked Dna2?”. The second is “Where is Dna2 localized in plant cells?”
Methodology
To determine what would happen to plants that lacked Dna2, knockout plants that lacked Dna2 were created. A. thaliana plants that were heterozygous for a TDNA insert in the DNA2 gene were ordered from the Arabidopsis Biological Resource Center. These heterozygous plants had one functioning DNA2 gene and one defective gene. These plants were selfcrossed to theoretically obtain homozygous, heterozygous and wildtype seeds. These seeds were grown next to wildtype seeds so that phenotypic differences could be observed. Plant genotype was determined by extracting DNA from plant leaves and using a polymerase chain reaction (PCR) to scan for inserts in the DNA2 gene.
In order to determine the location of Dna2 in the cell, Dna2 proteins were tagged with Yellow Fluorescence Protein (YFP) and viewed under a confocal microscope. To do this, a DNA fragment which encoded Dna2 was isolated from Arabidopsis DNA using PCR. This DNA fragment was then inserted into a pENTR/D TOPO bacterial vector and cloned. This recombinant vector (containing DNA2) was mixed with another vector, called pK7YWG2, which contained YFP. The DNA2 gene from the first plasmid was transferred to the second plasmid using homologous recombination. This placed the DNA2 gene in the same reading frame as YFP, creating a chimeric DNA2/YFP gene. This new vector was transformed into Agrobacterium tumefaciens. Agrobacterium is a unique bacteria that is able to infect plant cells and insert its own DNA into them. Agrobacterium was used to infect Nicotiana benthamiana leaves with the DNA2/YFP plasmid. These leaves were treated with Mitotracker dye and DAPI nucleic acid stain and then viewed under a confocal microscope.
Results
Arabidopsis plants homozygous for DNA2 knockout genes could not be obtained. The zygosity of 24 plants was determined by PCR. 15 were heterozygous for the knockout gene and 9 were wild type. This suggests that the knockout gene was lethal when homozygous, suggesting that Dna2 plays a vital role in Arabidopsis.
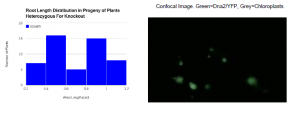
No phenotypic differences were observed in heterozygous knockout plants except for root length in weekold plants. Root length distribution in wild type plants created a bellshaped curve. In seedlings which were the offspring of heterozygous plants a curve with two separate peaks was created (see figure). One peak represented the root length of wild type plants, while the other peak represented heterozygous plants.
Confocal microscope analysis of leaves transfected with hybrid Dna2/YFP proteins suggests that Dna2 is localized to the chloroplast, possibly the mitochondria, and not the nucleus. YFP was observed in the same parts of the cell as chloroplasts, clearly showing that Dna2 must be in the chloroplasts (see figure). No YFP was seen in the nucleus. It is unclear whether or not Dna2 is found in the mitochondria. The mitotracker dye used here was originally meant for staining animal mitochondria, but it didn’t stain plant mitochondria.
Discussion
Since plants lacking Dna2 were never found, Dna2 must be vital for life in Arabidopsis. Dna2 is expected to function as both a helicase and endonuclease. Since plants cannot survive without Dna2 it appears that it is the only protein in plants that can fulfill at least one of those functions. Another protein called Twinkle is known to act as a helicase in plant organelles, so Dna2 is likely vital to plant cells because of its unique endonuclease capabilities.
Heterozygous knockout plants showed short root length during germination, suggesting that Dna2 is especially vital at that time. In order to test if Dna2 is still necessary in mature plants our lab is planning on using RNAi to knock out DNA2 expression in adult plants.
Transient transfection of Nicotiana benthamiana with Dna2/YFP has shown that Dna2 is localized to plant chloroplasts. However, in order to confirm these results, our lab has already begun permanent transfection of Arabidopsis plants with Dna2/YFP using floral dip methodology. Permanent transfection causes more cells to express Dna2/YFP and will give much better microscope images. Our lab is also permanently transfecting plants with Green Fluorescence proteintagged mitochondria. This will allow us to determine whether or not Dna2 is localized to mitochondria.