Jeffery Barrow, Physiology and Developmental Biology
My laboratory studies the role of Wnt signaling in regulating important events during embryogenesis. In order to better understand the function of this pathway, we study mouse mutants that lack the Porcupine (Porcn) gene which encodes a protein that is required for the secretion of all Wnt molecules. Prior to obtaining the MEG grant entitled: The role of Wnt signaling in normal muscle development and disease, we had demonstrated that mice lacking Porcn activity in the ventral limb ectoderm exhibit a thinning of the ventral limb ectoderm, loss of epidermal appendages and loss of muscle and tendons in the underlying mesoderm. The purpose of this grant was to characterize the basis of muscle and tendon loss in the Porcn; Msx2Cre mutants as well as to determine whether clonal mutations in the Porcn gene were the basis for ectodermal and muscular defects in individuals with Poland Syndrome. I will summarize the progress and accomplishment of each of the aims in the proposal.
Specific aim 1: We will test the hypothesis that the Wnts are the ectodermal signal that induces tendons of the limb.
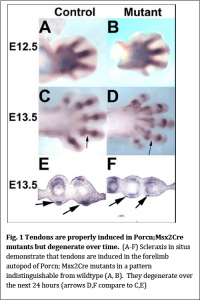
Previous work has demonstrated that signals from the limb ectoderm are required for the induction of mesodermal tendons. Other studies have demonstrated tendons are required for the presence of muscle. We therefore reasoned that the primary defect in Porcn; Msx2Cre mutants (which lack ventral limb musculature) could be that ventral tendons are not induced which then prevents that formation of ventral musculature. We performed in situ hybridization using Scleraxis as a marker for tendons and determined that at E12.5 (the stage at which tendons are forming in the forelimb autopod (paw), tendons form in the mutants similar to what is observed in wildtype (Fig.1). Twenty-‐four hours later at E13.5 tendons degrade (Fig. 1). Therefore Wnts are not the ectodermal signal required for tendon induction. Their degeneration is likely secondary to the absence of ventral musculature.
Specific aim 2: We will test whether muscle loss in the ventral limb, perineum, ventral body wall in Porcn; Msx2Cre mutants are due to loss of SF/cMet, SDF1/CXCR4 or TCF4 expression.
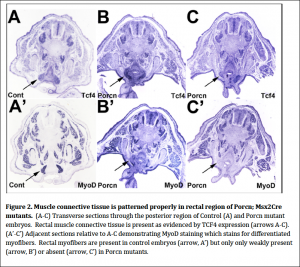
We examined the expression of two secreted factors and their respective receptors in wildtype and Porcn; Msx2Cre mutants to determine if these signaling systems which are required for normal migration of migrating myoblasts were disrupted in the mutants. We therefore performed in situ hybridization of SF and its receptor c-‐Met, SDF1 and its receptor CXCR4 and found no discernible differences between mutants and controls. We also examined the expression of TCF, a transcription factor expressed in mesodermal connective tissue that acts as a scaffold for migrating myoblasts and assists them in patterning the various muscles. We found that TCF4 was expressed in muscle connective tissue just as in controls. Unlike controls, however, myoblasts are not arriving at the connective tissue (Fig. 2). This result demonstrates that there are no defects in the presence and pattern of the muscle connective tissue. Therefore the myoblasts are being lost prior to arriving at the muscle connective tissue targets.
Specific Aim 3 We will test the hypothesis that mesodermal sources of Wnt signals are responsible for patterning centrally localized muscles in the limb.
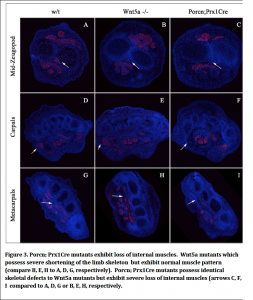
We have demonstrated that when Porcn is removed from the ventral limb ectoderm there is a loss of superficial ventral muscles and tendons. We reasoned that removing Porcn function mesodermally would affect the formation of more central muscles. We therefore generated Porcn; Prx1Cre mutants where Prx1Cre removes Porcn in the early limb mesoderm cells. We found that indeed Porcn:Prx1Cre mutants exhibit loss of centrally localized muscle (Fig. 3). Therefore secreted Wnt signals regardless of whether or their source play a general role in assisting migrating myoblasts from their origin in the somite to their final destination in the limb. We reasoned that it was possible that skeletal defects that exist in Porcn Prx1Cre mutants might have been secondarily responsible for the muscle loss that we observed. We therefore compared the muscle phenotype with Wnt5a mutants that have identical skeletal defects to Porcn; Prx1Cre and found that Wnt5a mutants exhibit normal muscle pattern demonstrating the muscle defects observed in the Porcn mutants are not secondary to skeletal defects.
Specific Aim 4 We will test the hypothesis that Poland Syndrome anomalies are due to clonal loss of Porcn in the affected ectoderm PS patients.
Individuals with Poland Syndrome (PS) exhibit thinning of pectoral epidermis, loss of epidermal appendages (i.e., hair and nipples) and absence of of underlying muscle. These are precisely the defects that we observe in mice lacking Porcn activity in the ectoderm. We established a collaboration with Dr. Paul Cerderna, MD at the University of Michigan as one who could send us epidermal samples from normal and affected skin of PS patients so that with the assistance of Dr. Keone Kauwe we might determine whether PS was due to clonal loss of Porcn in the epidermis of PS patients. For reasons that are not entirely clear we were never able to obtain affected tissue from Dr. Cederna.
Future Directions
My present graduate student, Tanya Milburn is continuing to work on this project. She is examining the hypothesis that ectodermal Wnts are required for proliferation and/or survival of myoblasts as they migrate to the destinations in the limb.
Academic deliverables from this study
Much of this work was published as an abstract and presented at the Southwest Regional Meeting of the Society of Developmental Biology in Salt Lake City in 2013. Jordan Huber, an undergraduate in the lab who did much of the work detailed above, presented this work (Huber, J, Smith, A.P., Barrott, J. J., Murtaugh, L. C., Barrow, J.R. (2013) Ectodermal Wnt signaling is required for muscle patterning. Society of Developmental Biology Southwest Regional Meeting, Salt Lake City).
Since Fall 2013, Tanya Milburn has taken over the project. Upon completion of her work we anticipate publishing this work in a reputable developmental journal (i.e., Development, Developmental Biology or Developmental Dynamics).
I mentored 4 outstanding students with the mentoring grant. Jordan Huber, an undergraduate project leader on the project, graduated Summa Cum Laude in April of 2013. He is now 2nd year student at University of Utah School of Medicine. Tanya Milburn took over the project in fall of 2013 is now in her 2nd year of her masters thesis in my laboratory. She has had two undergraduates that have assisted her in her work: Jared Holley-‐graduated BYU PDBio April of 2014 and is presently a tutor at Altius and is applying to medical school; Nathan Kmetzsch will graduate April 2015 has several medical school acceptances.
The MEG grant funds were used to pay for animal housing costs and reagents for the experiments (in situ supplies, histology costs, antibodies, confocal microscopy costs). In addition they paid for registration and poster costs for Jordan Huber. This MEG grant has provided enormous support to my laboratory and has provided critical funds to help collect important data in wrapping up an important study in the laboratory.