Sara Mason and Dr. Eric Wilson Microbiology and Molecular Biology
Introduction:
Chemokines play a well-characterized and significant role in the immune system as chemoattractants for white blood cells. Some chemokines have also been shown to have antimicrobial properties in addition to their chemoattractant functions. CCL28 is a chemokine that aids in the recruitment of antibody secreting cells to mucosal surfaces of the body, especially in the large intestine and lactating mammary glands.1 Recent research has shown that, in addition to recruiting lymphocytes, CCL28 also exhibits antimicrobial activity against gram positive and gram negative bacteria, and antifungal activity.2
In this study we wanted to explore the role of CCL28 as an antimicrobial peptide in the large intestine. Our lab has shown that when recombinant CCL28 is added to isolated intestinal bacteria harvested from a mouse, the CCL28 sticks to the bacteria and is detectable through flow cytometry. We were interested if we could detect endogenous CCL28 bound to commensal bacteria in the large intestine, rather than just the recombinant CCL28 we added. Methodology: Two methods were used to detect endogenous CCL28: a flow cytometric biniding assay and enzyme-linked immunosorbent assay (ELISA).
Binding Assay:
Bacteria was extracted from the large intestine of an adult mouse and suspended in 1 ml PBS containing .1% BSA. Ten μl of this bacteria was added to a five ml tube containing 200 μl of PBS containing .1% BSA and10 μl biotinylated anti-mouse CCL28 antibody diluted 1:500. The samples were incubated on ice for 1 hour. The tubes were incubated on ice for 1 hour and then washed. Then the samples were resuspended in 100 μl PBS and incubated with 10 μl streptavidin conjugated with the fluorochrome allophycocyanin (APC) for 30 minutes on ice. The samples were washed again, and then resuspended in 150 μl PBS and read in a flow cytometer.
ELISA:
ELISA plates were incubated at room temperature (RT) overnight with 100 μL polyclonal anti-mouse CCL28 antibody diluted 1:500 in PBS. Plates were washed three times and then were blocked with PBS containing 1% (w/v) BSA for 1 hour at RT. Bacteria was extracted from the large intestine of an adult mouse and suspended in 200 μl PBS containing 1% (w/v) BSA. The plates were washed three times and then the bacteria samples were loaded into the top wells of the ELISA plate and serially diluted them down the wells in the same diluent and incubated at RT for 2 hours. Wells were again washed three times and incubated with 200 μl of biotinylated anti-mouse CCL28 antibody diluted 1:250 at RT for 2 hours. The plates were washed three times again and then incubated for 30 min with streptavidin conjugated to horseradish peroxidase at RT. Wells were washed three times and then incubated with 100 μl of TMB color substrate for 1 hour at RT. Then 50 μl .18 M H2SO4 was to each well and the plates were read at absorbance 450 nm.
Results:
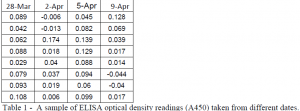
Results from the binding assays (not shown) were extremely inconsistent. Our assay was not sensitive enough to reliably detect such low levels of CCL28 found in the large intestine. ELISA results occasionally showed dilutions, but rarely. There was too much background interference to get a reliable reading, let alone consistent ones.
Discussion:
The large intestine is a sight of great activity for the immune system and the digestive system. Due to the preponderance of bacteria, enzymes, proteins, and the large intestine contents, there is inevitably a lot of background in these assays. Our assays were not sensitive enough to bypass this fact. While we did run into these troubles, we were able to conclude that there is endogenous CCL28 in the large intestine that can be detected. This will lead to further research exploring whether or not the presence of CCL28 in the large intestine has an impact on the bacterial composition of an individuals intestinal microflora.
References
- Morteau, Oliver, Craig Gerard, et al. “Journal of Immunology.” Journal of Immunology. 181 (2008): 6309-6315. Print.
- Heishima, Kunio, Haruo Ohtani, et al. “Journal of Immunology.” Journal of Immunology. 170 (2008): 1452-1461. Print.
