Christian Boekweg, Rachel Nettles, and Dr. Samuel St. Clair, Plant and Wildlife Science
In the aspen forests on the mountains of Utah, a shift is occurring in the ecological community. The aspen stands have, over time, been invaded by multiple conifer species. The composition of these forests is shifting from a pure aspen stand to a pure conifer stand (Calder & St. Clair 2012). Recent findings show that there exists in conifer-aspen ecosystems a direct facilitative relationship between aspen and conifer. While there have been multiple studies investigating the facilitative interactions of conifer-conifer stands, at present, little or no data is available regarding facilitation and seedling establishment in aspen dominated and aspen-conifer mixed stands. In this study we will look at the effect of temperature and water availability on the germination of four mountainous conifer species and aspen. These results can then be compared against the existing water potentials that are prevalent in aspen, and aspen-conifer stands thus allowing us to predict the probability of successful germination of conifer seedlings in the areas of question.
We attempted to gain insight into the facilitation and recruitment of conifer seeds by aspen trees by examining the environmental conditions that influence the germination of conifer seeds to see if aspen trees create hospitable environments for conifer seeds. The seed types we used were Ponderosa Pine (Pinus ponderosa), Engelmann Spruce (Picea engelmannii), Douglas Fir (Psudotsuga mensiesii), Subalpine Fir (Abies lasiocarpa), and Quaking Aspen (Populus tremuloides). The three factors we examined were water availability, temperature, and time.
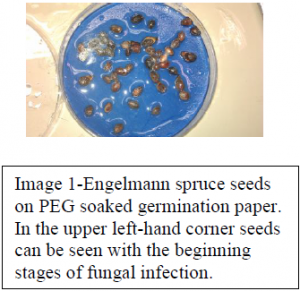
In order to account for variations in temperature we used incubators pre-set for a range of temperatures starting at 0 degrees C to 25 degrees C, with increments at 5 degrees C, 10 degrees C, and 15 degrees C. The effects on seeds from 20-25 degrees C has been shown to be minimal, thus we opted to only run a set at 25 degrees C. To account for variations in water availability seeds were placed in Petri dishes atop germination blotters purchased from Anchor Paper, St. Paul, Minnesota USA, that had been completely saturated with one of five PEG (Polyethylene Glycol) solutions calibrated to deliver the following water potentials respectively: 0 MPa, -0.5 MPa, -1.0 MPa., -1.5 MPa, and -2 MPa. The procedures followed in mixing PEG and water to achieve these water potentials were set forth by Michele and Kaufmann in their paper in 1972. (Image 1) In order to maintain seed integrity we followed the current protocol placing our dishes in sealed plastic sleeves, only removing them during seed checks. Seeds were checked for germination on days 2, 4, 7, 11, 14, 21, and 28. Upon reaching day 28 seeds were to be transferred into a free water environment for an additional 28 days to check for seedling viability in an effort to ensure that the lack of germination we observed was indeed due to the water potential or temperature that the seeds experienced rather than from bad (already dead) seeds. Experimental sets run in quadruplicate to ensure validity in results with all water potentials being accounted for in every seed type, and temperature. All seed types being present in all temperatures and water potentials, and every temperature having all seed types in all water potentials.
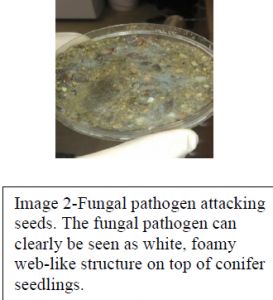
On day 14 of our experiment we observed signs of contamination from Fusarium, a potentially lethal pathogen to seeds (Image 2). Fusarium is a filamentous fungi commonly found in soil and has been the subject of studies conducted at BYU by other students and professors. The unforeseen consequence of placing our seeds in an environment shared with Fusarium was the risk of exposure. This pathogen attacks and kills seeds that lay in soil seed bank prior to seed germination. Due to cross contamination our seeds showed signs of a Fusarium infection and were at risk. By day 21 we observed severe contamination and widespread seedling mortality in both our control and experimental groups. With the majority of our seeds dead or being attacked by the pathogen our experiment and the results we had hoped for were compromised. Due to the nature of our methods we were unable to conduct a duplicate experiment due to the restraints of both cost and time.
While we were unable to achieve the results we had hoped for, and our questions concerning the aspen-conifer facilitation remain unanswered, we took the knowledge we gained concerning the contamination issues in the incubator lab and applied it accordingly. After working extensively with Dr. Phil Allen we put in place new protocols that will greatly reduce the rates of infection and cross contamination in incubators. The protocols we implemented are mandatory sterilization of containers via autoclaving, new containers to place bags in (creating a 3rd barrier to the pathogens being studied by other students), and routine, schedule cleaning and sterilization of the incubators themselves.
In conclusion it is our hope that with these new protocols and procedures cross contamination in the incubators can be eliminated, thus preserving the integrity of our future experiments and the experiments of others. Also it is our hope that with the start of a new semester we might be able to repeat this experiment (devoid of contamination) and gain insight into this important aspect of Utah’s forest ecosystem.
References
- Calder W.J., and St.Clair S.B. (2012) Facilitation drives mortality patterns on succession gradients of aspen-conifer forests. Ecosphere 3 (6): 57.
- Michel, B.E., and Kaufmann, M.R. (1972) The osmotic potential of polyethylene glycol 6000. Plant Physiology 51, 914-916.