Benjamin Fisher and Dr. Laura Bridgewater, Molecular Biology
Osteoarthritis (OA) is a disease that affects 26 million of Americans, yet its cause it not understood.1 It is a degenerative disease that affects cartilage at joints, and there are several risk factors that give people a higher probability of developing OA; these risk factors include acute joint injury, obesity, and family history of OA. Scientists are still trying to discover the genetic cause of OA, but no one has been successful at finding the link. In our research, we attempted to use 2 mouse models with genetic mutations to study the proteins involved in OA. Dmm (Disproportionate Micromelia) and Sedc (Spondyloepiphyseal Dysplasia Congenita) mice have mutations in the Collagen II gene that predisposes them to develop OA.2 We focused first on Dmm mice, because their symptoms are more severe, including dwarfism and abnormal cartilage, so they are easier to study. Sedc mice provide more promise for a link to human OA, because their symptoms are more similar. Both mice have a mutation that prevents the Collagen II from folding properly to create the triple-helical structure, so it affects the integrity of the cartilage.
However, we believe the Unfolded Protein Response (UPR) is also activated and contributes to the development of OA. The UPR is the cell’s response to excess proteins accumulating in the Endoplasmic Reticulum (ER). The ER is responsible for folding proteins, and a back-log in the ER indicates there are excess unfolded proteins, so the UPR decreases protein synthesis of most proteins and increases production of chaperone proteins. These chaperones help fold and transport proteins out of the cell. UPR also causes inflammation, and both of these pathways help to restore homeostasis. If homeostasis isn’t achieved, the UPR can eventually trigger apoptosis (cell death).3 Since Dmm and Sedc mice have mutations that always cause them to produce abnormal Collagen II proteins which cannot fold properly, they are an excellent model to study the effects of UPR.
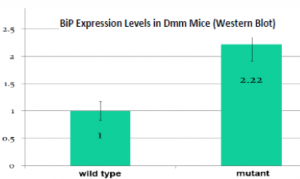
In our initial studies of the Dmm mice, we were able to show that BiP (one of the first protein chaperones activated by the UPR) had twice the expression levels in newborn mutant mice (D/+) than wild-type mice (+/+). We dissected and took knee cartilage from newborn mice, homogenized the tissue, and analyzed using Western Blots (see figure below). Our research goal was to take the information we’d learned about Dmm mice and expand upon it; we planned to isolate chondrocytes from the knee cartilage, culture them, and analyze the proteins in the cells to show definitively that we had actually induced the UPR.
Unfortunately, we were unable to harvest and isolate enough chondrocytes to allow us to analyze them, so we chose another option. Using Real-Rime PCR, another student cultured HEK 293 cells and treated them with tunicamycin (a chemical used to induce UPR). That created a positive control to show that the chaperone proteins were being expressed at a higher level in UPR, giving us confidence in claiming that the elevated levels we saw in our mice could be attributed to the UPR as well. Unfortunately, this experiment didn’t yield the results we hoped. We have dissected, isolated, and homogonized knee cartilage from newborn and 2-month mice, both wild-type and mutant, to analyze using Western Blots. While BiP has higher expression levels in Dmm mice, we’ve been unable to show increased levels of any of the other chaperone proteins, including CHOP and the phosphorylated forms of IRE1alpha and eIF2alpha. We also did the same process with Sedc mice and have found no significant increase in protein expression levels. We’ve been having trouble with our antibodies, so we’re running a few more samples to confirm those results, but at this point we have no additional evidence to show that the UPR is the cause of Osteoarthritis.4
References
- Helmick, C., Felson, D., Lawrence, R., Gabriel, S., et all. Estimates of the Prevalence of Arthritis and Other Rheumatic conditions in the United States. Arthritis & Rheumatism 58(1), 15-25. 2008.
- Kannu P, Bateman JF, Randle S, Cowie S, du Sart D, McGrath S, Edwards M, Savarirayan R. Premature arthritis is a distinct type II collagen phenotype. Arthritis Rheum. 2010 May; 62(5):1421-30. PubMed PMID: 20131279.
- Seegmiller RE, Bomsta BD, Bridgewater LC, Niederhauser CM, Montaño C, Sudweeks S, Eyre DR, Fernandes RJ. The heterozygous disproportionate micromelia (dmm) mouse: morphological changes in fetal cartilage precede postnatal dwarfism and compared with lethal homozygotes can explain the mild phenotype. J Histochem Cytochem. 2008 Nov; 56(11):1003-11. Epub 2008 Aug 4. PubMed PMID: 18678883; PubMed Central PMCID: PMC2569899
- Special thanks to Brigham Young University’s Office of Research and Creative Activities (ORCA) for funding this research, my fellow lab researchers for their excellent work, and Dr. Laura Bridgewater for her mentorship and expertise. Working in her lab has been a wonderful experience and enriched my educational experience at BYU.