Susan Eldredge and Dr. Craig E. Coleman, Botany and Range Science
Zein proteins represent the prolamin (alcohol soluble) fraction of maize storage proteins. Zeins constitute approximately 50 percent of the total seed protein in maize, and therefore greatly affect the overall amino acid balance of the maize grain (1).
Four different zein subunits (á, â, ã, and ä) aggregate in maize endosperm to form maize protein bodies. The different subunits are spatially arranged within the protein bodies so that á-zein and äzein form a spherical core which is enclosed by a shell of ã-zein and â-zein (2, 3). Transgenic research using tobacco has shown that different zein subunits interact during protein body formation, increasing stability of the subunits thus making zein aggregation and accumulation possible (4, 5).
The current experiment was designed to investigate the interactions between the â-zein and ã-zein subunits in transgenic tobacco. The tobacco seeds used in this study had been previously harvested from plants that had been transformed with combinations of â-zein and ã-zein genes from maize. During preliminary work the seeds were embedded in LR White resin in preparation for thin sectioning.
This study commenced by sectioning the transformed tobacco seeds and collecting silver sections on nickel grids. In order to identify and differentiate zein subunits the grids were soaked in zein antiserum collected from rabbit (for ã-zein) or from rat (for â-zein). Primary-labeled samples were then soaked in a solution containing secondary antibodies conjugated to gold particles of diameters specific to the subunit type. Samples were the post-stained with lead citrate and uranyl acetate and viewed under the transmission electron microscope.
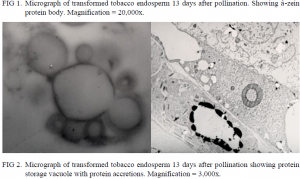
Despite multiple efforts in immunostaining, efforts to label the zein subunits with gold-conjugated antibodies were unsuccessful. Therefore, it was impossible to obtain conclusive data about the interaction between â- and ã-zein subunits during protein body formation. However, â-zein protein bodies were identified by their characteristic rosette shape in seeds that had been transformed by a combination of â- and ã-zein genes (Fig. 1) (6). Also identifiable in the transformed tobacco were protein storage vacuoles with electron dense accretions of protein bodies (Fig. 2), but without immunostaining, it is impossible to classify the protein as zein, or even to distinguish it as tobacco protein or maize protein (7).
Efforts to identify protein bodies in transgenic tobacco through immunolocalization continue. The techniques used in such experiments involve many variables and must be completed with precision. However, as the techniques are refined, it will be possible to obtain more conclusive data.
References
- Shotwell, M. A., and Larkins, B. A. (1989). The biochemistry and molecular biology of seen storage proteins. In: A. Marcus (ed.) The Biochemistry of Plants: a Comprehensive Treatise, vol. 15. New York: Academic Press.
- Lending, C. R., and Larkins, B. A. (1989). Changes in the zein composition of protein bodies during maize endosperm development. The Plant Cell 1: 1011-1023.
- Esen, A., and Stetler, D. A. (1992). Immunocytochemical localization of ä-zein in the protein bodies of maize endosperm cells. American Journal of Botany 79: 243-248.
- Coleman, C. E., Hermann, E. M., Takasaki, K., and Larkins, B. A. (1996). The maize ã-zein sequesters á-zein and stabilizes its accumulation in protein bodies of transgenic tobacco endosperm. The Plant Cell 8: 2335-2345.
- Bagga, S., Adams, H. P., Rodriguez, F. D., Kemp, J. D., Sengupta-Gopalan, C. (1997). Coexpression of the maize ä-zein in endoplasmic reticulum-derived protein bodies formed by â-zein. The Plant Cell 9: 1683-1696.
- Bagga, S., Adams, H. P., Kemp, J. D., and Sengupta-Gopalan, C. (1995). Accumulation of 15-kilodalton zein in novel protein bodies in transgenic tobacco. Plant Physiol. 107: 13-23.
- Nielsen, N. C., Bassüner, R., and Beaman, T. (1997). The biochemistry and cell biology of embryo storage proteins. In B. A. Larkins and I. K. Vasil (eds.) Cellular and Molecular Biology of Plant Seed Development. Dordrecht, Netherlands: Kluwer Academic Publishers, 151-200.