Michael F. Erickson, Department of Zoology
Introduction
Recent studies have shown that the cytokines interleukin-6 (IL-6) and tumor necrosis factor (TNF) are produced by cells found in the zonaglomerulosa of the rat adrenal cortex (I ,2). These cytokines modify adrenal hormone release gland. Specifically, IL-6 stimulates the release of glucocorticoids from the zona fasciculata/reticularis (3) and TNF inhibits the release of aldosterone from the zona glomerulosa (4). It is of interest that during times of traumatic injury or septic shock the serum and tissue levels of these cytokines increase. This increased cytokine level is partly responsible for the lethal effects of septic shock (including severe hypotension) since IL-6 and TNF decrease vascular tone and modify the release of aldosterone and glucocorticoids. Aldosterone and glucocorti· colds are important hormones in maintaining the integrity of the circulatory system. Therefore, fluctuations in the serum levels of these steroids due to IL-6 and TNF weaken the body’s ability to recover from the cardiovascular collapse that occurs during septic shock and traumatic injury.
An important next step is to determine the cell type(s) within the rat zona glomerulosa that is (are) releasing IL-6. It is possible that the cells that release aldosterone are also releasing IL-6. This is called autocrine control, where the release of one substance from one cell type modifies the release of another substance from the same cell type. Conversely, it is possible that a cell type that is distinct from the aldosterone-releasing cells is releasing IL-6. This is called paracrine control, where the release of a substance from one cell type modifies the release of a substance from a different cell type. There is also the possibilities that IL-6 and TNF are being secreted by some cell type found in the blood or connective tissues (e.g. wandering macrophages, fibroblasts or vascular endothelial cells) and therefore the release of IL-6 from adrenal tissues is an artifact of cell culture. The aim of this research was to determine the cell type(s) within the zone glomerulosa that secretes IL-6. This is important for two reasons. First, to determine whether aldosterone release is under autocrine or paracrine control. Second, to dismiss the possibility that IL-6 is being released by circulating macrophages, fibroblasts or other connective tissue cells and thus is an artifact of the cell culture.
Procedures
Female Sprague-Dawley (SASCO, Omaha, NE) rats were decapitated and the adrenal glands removed. In a laminar flow hood, fat and connec· tive tissue were removed. The adrenals were then transferred to a Petri dish containing 10 ml fresh complete RPM! 1640 culture medium (5), finely diced, and washed with the culture medium to remove trapped blood. They were then placed in a centrifuge tube containing I 0 ml complete RPM! and 20 mg collagenase (Sigma, St Louis, MO) and incubated at 3r C for one hour. The adrenal tissue was then dispersed with a pipette until a suspension of single cells were obtained. The dispersed cells were diluted to 500,000 cells/ml, 50 ul (25,000 cells) added to each well of an 8-well chamber slide (Baxter Scientific, Salt Lake City, UT) containing 200 pi of complete RPM! 1640 medium, and cultured 5-8 days at 37o C under an atmosphere of 95% room air, 5% carbon dioxide. The old culture medium was then removed and replaced with 200 pL of serum-free RPM! medium (5). This medium was then removed and replaced with ZOO pL of serum-free RPM! 1640 medium containing either vehicle (Z pL of serum-free RPM! medium) or 10 ng/mL lipopolysaccharide (LPS) (Sigma, StLouis, MO). LPS Increases IL-6 release from adrenal cells (1,Z). The cells were then incubated for Z hours at 37′ C under an atmosphere of 95% air. 5% carbon dioxide. The Incubation medium was then removed and the cells fixed by two washes with 4o C basic salt solution (BSS) (5), followed by a five minute incubation with ZOO pL of 4oC 4% paraformaldehyde (Sigma) in BSS. This was followed by two more washes with 4o C BSS. The cells were then permeabilized by subjecting them for five minutes (all steps after this point are at room temperature) to ZOO pL of BSS containing 1% bovine serum albumin (BSABSS). This medium was then replaced with ZOO pL of 0.1% Saponin (Sigma) in BSA-BSS(BSS-Saponin). This medium was rapidly removed. and the cells then incubated for five minutes in an additional ZOO pL of BSS-Saponln. This medium was then replaced with a medium containing the first antibody (13.4 pg/mL of rabbit anti-rat IL-6 or a 1/100 dilution of rabbit antirat p450 side chain cleavage enzyme) (ZOO pL In 0.1% BSS-Saponin) and the cells incubated In this medium for 75 minutes. The first antibody was removed. and the cells were washed three times with BSS-Saponln. A peroxidase suppressor (ZOO pL) (Pierce, Rockford, IL) was then added for 30 minutes to inhibit endogenous peroxidase activity associated with normal adrenal cells. The cells were then washed three times with BSS saponin. The second antibody (111000 dilution of donkey antirabbit IgG conjugated to horseradish peroxidase) (Pierce). was then added In ZOO pL of BSS-Saponin containing 1% rat serum and allowed to Incubate for 45 minutes. After removal of the second antibody, the cells were then rinsed three times with BSS-Saponin and a metal enhanced DAB detection kit (Pierce) was used to visualize the horseradish peroxidase conjugated second antibody. Using the DAB detection kit. a small black granule inside the cell was positive evidence for the presence of horseradish peroxidase. The cells were then observed under a light microscope, and the percentage of cells with granules were noted along with the number of granules per cell.
Results
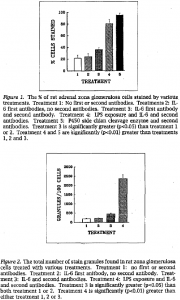
The above procedures were carried out six times. The average of these six trials is presented in Figures 1 and Z. As a control for nonspecific labeling, some cells were treated with the above described procedure minus the first and second antibodies. Under these conditions, Zl.4% of the cells had granules with an average of 9.3 granules per cell for a total of 199 granules per 100 cells. A second control involved the nonspecific labeling of cells that had been exposed to the above treatment minus the second antibody. This treatment resulted In Z4% of the cells with granules and 9.0 granules per cell for a total of Z16 granules per 100 cells. There was no statistical difference between these two control groups. The basal IL-6 group consisted of the cells treated with both antibodies (IL-6 and second antibody). but not treated with LPS. In this treatment, 35.5% of the cells had granules with an average of 13.0 granules per cell for a total of 416.5 granules per 100 cells. This treatment resulted in a significant (p<0.05) increase in the staining of the cells when compared on a percentage basis, granules per cell basis, or total number of granules per 100 cells basis. In the adrenal cells exposed to LPS and IL-6 and second antibodies, 80.5% of the cells had granules with an average of Z9.4 granules per cell for a total of Z367 granules per 100 cell. This group was significantly Increased (p<0.01) when compared to both the control and basal treatment groups. The final treatment represents cells that were stained with anti-P450 side chain cleavage enzyme antibody. In this group. a large percentage of the Intracellular membranes were stained, and therefore, it was very difficult to identify individual granules. However, this stain resulted in over 90% of the cells in the culture being stained.
Discussion
The immunostaining resulted in over 80% of the cells being labeled as positive for secreting IL-6 following exposed to LPS. Because over 90% cells in the rat zona glomerulosa culture also stained for P450 side chain cleavage enzyme, an enzyme found in cells that secrete steroid hormones, we can be confident that the steroid-secreting cells of the zona glomerulosa are also secreting IL-6. Because IL-6 affects the function of the rat adrenal cortex, the effect of IL-6 on the secretion of aldosterone, a steroid hormone, is probably an example of autocrine control. Our results also strengthen the hypothesis that the majority of the IL-6 in the adrenal is being released from the zona glomerulosa cells and not from wandering macrophages, fibroblasts or vascular endothelial cells. This conclusion is substantiated because of the fact that macrophages, fibroblasts and endothelial cells, cells that do not stain for P450 side chain cleavage enzyme, make up only a small percentage of the cells in the culture (less than 10% not 80%). However, we cannot exclude the possibility that some of the IL-6 present in the rat zona glomerulosa cultures may come from cells that do not secrete steroids. The increase in IL-6 staining following LPS exposure correlates well with data that demonstrate that LPS increases IL-6 release from rat adrenal zona glomerulosa cells (1,2). ACTH, a peptide hormone released from the anterior pituitary during stress also increases adrenal IL- 6 release. Furthermore, IL-6 affects the function of the adrenal gland. Therefore, LPS and ACTH may affect adrenal function in part by increasing the production of IL-6 by the adrenal gland. This may be of particular importance during stress and bacterial infections.
References
- A.M. Judd and R.M. MacLeod. “Differential Release of Tumor Necrosis Factor and IL-6 From Adrenal Zona Glomerulosa Cells in Vitro.” Am. _1 Physiol. 1995. 268 (Endocrinol. Metab. 3 1): E114-E120.
- A.M. Judd and R.M. MacLeod. “Adrenocorticotropin Increases Interleukin-6 Release From Rat Adrenal Zona Glomerulosa Cells.” Endocrinology. 1992. 130:1245-1254.
- M.A. Salas, S.W. Evans, M.]. Levell, and ].T. Whicher. “lnterleukin-6 and ACTH Act Synergistically to Stimulate the Release of Corticosterone From Adrenal Gland Cells.” Clin, Exp. Immunol. 1990. 79:470-473.
- R. Natarajan, S. Ploszaj, R. Horton, and J, Nadler. “Tumor Necrosis Factor and Interleukin-1 are Potent Inhibitors of Angiotensin-II-Induced Aldosterone Synthesis.” Endocrinology. 1989. 125:3084-3089.
- A.M. Judd and R.M. MacLeod. “Thyrotropin-Releasing Hormone and LysineBradykinin Stimulate Arachidonate Liberation From Anterior Pituitary Cells Through Different Mechanisms.” Endocrinology. 1992. 131: 1251- 1260.