Holly Waddel and Dr. Clinton Whipple, Department of Biology
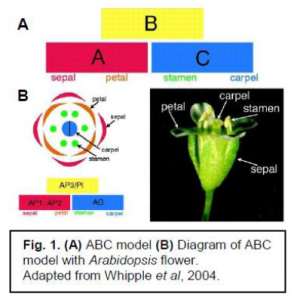
The evolution of floral development is caused by changes in gene function. The model for floral morphology is the ABC model of floral development (Coen & Meyerowits, 1991). In this model, A genes control sepals, A + B genes control petals, B + C genes control stamen, and C genes control carpels (See fig. 1). If B genes are removed, the resulting flower would contain only sepals and carpels, no petals or stamen.
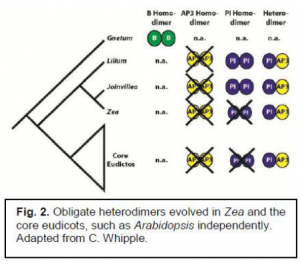
Joinvillea is a sister-group to the grasses. It shares with the grasses, and other distantly related flowering plants, a similar set of B-class subfamily genes, AP3-like genes and PI-like genes. The AP3-like gene produces a protein that must bind with the protein produced by the PI-like gene. This protein heterodimer can then function as a transcription factor for these Bclass genes. Having a functional AP3-like and PI-like gene means that B class genes can interact with the A and C genes to create a normal flower.
Joinvillea is unique, because it can bind two PI-like proteins into a homodimer (Winter et al, 2002). Zea, (corn, a grass) on the other hand, can only function as a PI-AP3 heterodimer. This means that obligate heterodimerization of the PI-AP3 genes evolved twice; once in the core eudicots, and once in the grasses (See fig. 2). This experiment was examining if this Joinvillea PI homodimer is a functional transcription factor, and if it has the same function as the PI-AP3 heterodimer during floral development.
We approached this project by taking the Joinvillea-PI gene and inserting it into a plasmid that can replicate in E. coli. This plasmid had the appropriate 10-OP promoter. This promoter is only functional where B-Class genes are normally transcribed, so it ensured our gene was read in the correct part of the plant. We then cut out the Joinvillea-PI and the 10-OP promoter and placed them in a new plasmid, pMLBART 27. This plasmid grew in both E. coli and Agrobacterium. Agrobacterium is a bacterium that infects plants by inserting a segment of DNA into the plant’s genome. We used this bacterium to insert our Joinvillea-PI gene into a core eudicot plant, Arabidopsis. Our Arabidopsis plants had non-functioning B-class genes.
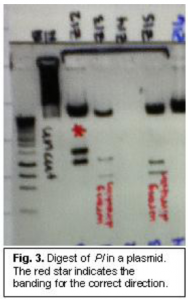
The main problems we had during this experiment were inserting our gene into the plasmid. It was necessary for the gene into plasmid in the correct direction. The plasmid read in a certain direction, and we needed to have the promoter read before the gene. The gene should have inserted correctly 50% of the time, but we had to insert the gene several dozen times before we found one correctly placed gene. To determine direction, we would extract the DNA plasmid from E. coli and use enzymes to digest the DNA into predictable segments. If the DNA pieces were sized correctly, we knew we had a correctly placed insert (see fig. 3).
Once we had a correctly placed Joinvillea-PI insert in our pMLBART 27 plasmid, we transformed it into the Agrobacterium. Using Agrobacterium to insert our gene into the plant Arabidopsis was the next challenge. Agrobacterium will transform Arabidopsis at an extremely low frequency (less than 1%). Our first attempt at transformation produced several thousand seeds but none of them were correctly transformed or the seeds didn’t survive. This set the project back several months, and the newly transformed seeds are still growing.
Our successful transformants are still too young to assess for recovery of the B-class gene. When this data is available, it will provide more support for a publication being prepared by Dr. Madelaine Bartlett for PLoS Biology or the Proceedings of the National Academy of Sciences. I will be included as a co-author on any of the publications resulting from this work.
References
- Coen ES, Meyerowitz EM. (1991). “The war of the whorls: genetic interaction controlling flower development. Nature. (353) 31-37.
- Whipple, C., Ciceri, P., Padilla, C. M., Ambrose, B. A., Bandong, S. L., Schmidt, R. J. (2004). “Conservation of Bclass floral homeotic gene function between maize and Arabidopsis.” Development (131) 6083-6091.
- Winter, K., Weiser, C., Kaufmann, K., Bohne, A., Kircher, C., Kanno, A., Saedler, H., Theiβen, G. (2002). “Evolution of Class B Floral Homeotic Proteins: Obligate Heterodimerization Originated from Homodimerization.” Molecular Biology and Evolution. 19(5) 587-596.