Dusten M. Macdonald and Dr. John D. Lamb, Chemistry and Biochemistry
Processing and storage of medium and high level activity liquid nuclear wastes (MLW/HLW) present a major challenge for governments around the world. Gamma radiation from 137Cs, as well as radiation from 90Sr and other radionuclides present in the waste, complicates the already troublesome disposal process (1, 2). Removal of such long-life radionuclides from MLW and HLW mixtures would allow for more affordable disposal of the partially decontaminated solutions, would lower the volume of high activity waste to be stored in geological formations, and/or possibly allow for transmutation of these radionuclides into non-radioactive elements (2). Much effort has been expended in searching for ways to effectively separate various radionuclides, particularly 137Cs, from nuclear waste mixtures. The most promising fruits of this research involve the use of macrocycles, relatively large cyclic ligands, to selectively form complexes with targeted nuclides and thereby facilitate their removal.
A suitable macrocycle for use in separating 137Cs from HLW or MLW must preferentially form complexes with Cs+ ions over other ions present in high quantities in waste mixtures, particularly K+ and Na+ (3). The macrocycle must also possess properties which allow it to be effectively incorporated into separation processes such as solvent extraction, supported liquid membrane, or polymer inclusion membrane systems. Recent studies have also shown that calix-crowns, specifically derivatives of calix[4]arene-crown-6 and calix[4]arene-bis-crown-6, may operate effectively in such solutions (2, 3, 4). The present challenge is to find the exact calix-crown derivatives which allow for optimum selectivity and may be effectively incorporated into separation systems.
Calixarene macrocycles generally consist of four or more phenyl groups linked together in a ring to form a cup shape. Calix-crown-ethers build on this motif by simply adding one or two crown ether loops stretching across the “cup” from one phenyl group to another. I used isoperibol titration calorimetry to find out how well didodecylcalix[4]arene-crown-6 (i.e. a calix-crown-ether containing four phenyl groups and a six-oxygen crown ether chain, substituted with two didodecyl groups) complexes alkali metal cations, including Na+, K+, Rb+ and Cs+. Tatiana Levitskaia, a graduate student working with Dr. John Lamb of the Chemistry Department, synthesized this calix-crown-ether.
Isoperibol titration calorimetry measures the temperature of a system as a function of titrant added, and provides a useful and accessible method for thermodynamic analysis.5 With a ligand solution as titrate and a metal salt solution as titrant, this technique allows determination of four related thermodynamic values ( H, G, S, and K) describing the complexation of the ligand and the metal ion. I titrated solutions of NaSCN, KSCN, and RbSCN into pure ligand (didodecylcalix[4]crown-6) solutions, using HPLC grade acetonitrile as the solvent in all of these solutions. I titrated CsSCN into a solution of ligand and KSCN, and used a competition scheme to calculate thermodynamic values for Cs+ complex formation. In order to establish precision, each experiment was performed between three and six times, and appropriate dilution runs were performed as each system requires. Calculations were made using the computer programs ENPLT, AVDIL, EQDH, and REACTIONS, currently installed on the Physics Department VAX computer.
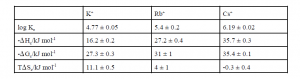
Thermodynamic values for the complexation of alkali metal cations with didodecylcalix[4]arenecrown- 6 obtained through this research are as follows:
Values for the complexation of Na+ with didodecylcalix[4]arene-crown-6 were not obtained because this complexation reaction does not produce sufficient heat to allow accurate measurements. These thermodynamic values confirmed that didodecylcalix[4]arene-crown-6 selectively forms complexes with Cs+ ions over other alkali metal ions present in nuclear waste mixtures. Understanding the complexing properties of calix-crown-6 derivatives will aid in applying these promising ionophores to nuclear waste clean-up technology. A more complete description of this research is available in reference 6.
References
- Blasius, E.; Nilles, K.-H. Radiochim. Acta 1984, 35, 173-182.
- Hill, C.; Dozol, J.-F.; Lamare, V.; Rouquette, H.; Eymard, S.; Tournois, B.; Vicens, J.; Asfari, Z.; Bressot, C.; Ungaro, R.; Casnati, A. J. Inclusion Phenom. Mol. Recognit. Chem. 1994, 19, 399-408.
- Haverlock, T. J.; Bonnesen, P. V.; Sachleben, R. A.; Moyer, B. A. Radiochim. Acta 1997, 76, 103-108.
- Asfari, Z.; Bressot, C.; Vicens, J.; Hill, C.; Dozol, J.-F.; Rouquette, H.; Eymard, S.; Lamare, V.; Tournois, B. Anal. Chem. 1995, 67, 3133-3139.
- Christensen, J. J.; Izatt, R. M. Calorimetric Techniques to Study Proton-Ligand and Metal-Ligand Interactions. Presented at MATO Summer School, Tabiano, Italy 1979.
- Macdonald, D. M. Calorimetric Investigation of the Complexation of Didodecylcalix[4]arene-crown-6 with Alkali Metal Cations in Acetonitrile. Honors Thesis, Brigham Young University, Provo, UT, September 1998.