Nathan Evanson and Dr. Daniel L. Simmons, Chemistry and Biochemistry
Cyclooxygenases (COXs) are enzymes that are responsible for the production of a potent class of biological signaling molecules called prostaglandins. Prostaglandins are involved in responses such as inflammation, contraction of smooth muscle, and regulation of water balance in the kidney. They are also found in greatly elevated amounts in some types of cancer. This elevation in prostaglandins is the result of increased production of the form of COX known as COX-2. Because COX-2 may play a role in the development of cancer, an understanding of how COX-2 is regulated is important in the understanding of cancer and may give insights that could lead to a cure.
In the chicken, COX-2 is highly up-regulated in rous sarcoma virus-induced cancer. The chicken is a convenient model for studying COX-2 regulation. In non-growing chicken cells, the gene for COX-2 is transcribed into RNA and prepared for production of protein, except that one of the introns (junk RNA that must be removed for protein to be made) is not removed. COX-2 mRNA containing the intron is retained in the nucleus of the cell, where it cannot be used to make protein. Treatment of the cells with things that cause them to divide rapidly or turn into cancer cells (such as rous sarcoma virus infection), causes the intron to be removed, allowing COX-2 protein to be made. Part of the intron and other parts of the RNA regulate the removal of the intron. The purpose of this study was to determine what part of the intron causes the intron to be removed correctly.
Deletion of a 500 nucleotide long piece of the COX-2 RNA largely eliminates splicing of the intron to the correct splice site (see Figure 1). This fragment alone is not sufficient to allow correct removal of the intron, however. Figure 2 shows the results of an experiment using a luciferase construct with luciferase intron 1 replaced by COX-2 intron 1. The vertical values on the graph indicate the activity of luciferase in the different constructs. The bar labeled pRSV-Luc is the positive control (luciferase without an intron). The bars labeled Beta-gal and pGEM-luc are negative controls that do not contain luciferase. The one labeled pLuc-I1 is another negative control that has the intron in luciferase (the intron will not be excised without another signal in the RNA). The other two are the experimental treatments, which showed that the 500 b.p. alone is not enough to allow correct splicing.
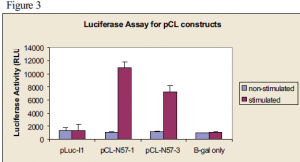
Insertion of part of this 500 nucleotide sequence into a gene made up of parts of luciferase and COX-2 appended to each other that allows a small amount of splicing appeared to suppress splicing (see figure 3). The bars labeled pCL-N57-1 represent the positive control and the ones labeled pCL-N57-3 represent the experiment. The lower luciferase activity in pCL-N57-3 indicates a possible suppression of splicing.
Conclusions
The expression COX-2, which is a protein that is highly induced in many types of cancer, is tightly regulated in chicken, by a novel regulatory system. In this system, the gene is activated upon neoplastic transformation of the cells being studied. Understanding of how chicken COX-2 is regulated at the post-transcriptional level in chicken may lead to new therapies for cancers in which the same signal transduction pathways are active.
Work that remains to be done includes identification of the exact signal that causes the intron to be removed and the protein to therefore be expressed and identification of the element(s) that are responsible causing the intron to be removed accurately.