Dr. Morris J. Robins and Dr. Theodore C. Jessop, Chemistry and Biochemistry
2-amino-6-alkoxypurineribosides are involved in clinical testing as anticancer co-drugs (1); however, current methods for the large scale synthesis of these compounds have proven extremely difficult. The improved synthesis of 2-amino-6-alkoxypurineribosides reported herein allows easy access to these medicinally important compounds.
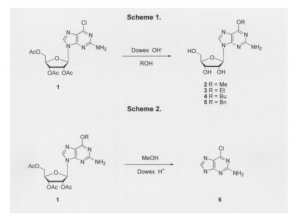
The novel reaction of this synthesis is summarized in Scheme 1. Compound 1, 9-(2,3,5-tri-O-acetylb- D-ribofuranosyl)-2-amino-6-chloropurine, was prepared from guanosine using established methods (2). The Dowex anion exchange resin was rinsed, suspended in alcohol and compound 1 added. Reaction times ranged from four hours for methyl alcohol to two days for butyl alcohol. Upon completion of the reaction, a small amount of cysteine was added to the mixture to quench any unreacted starting material. The mixture was then added to a column and eluted with an alcohol/acetic acid solution. Evaporation gave the 2-amino-6-alkoxypurineriboside (compounds 2, 3, 4, 5) in yields ranging from 60 to 99 percent.
Complications I encountered included conditioning of the Dowex OH- for use in solvents not miscible with water. In these cases, the Dowex was rinsed with copious amounts of the alcohol or conditioned via a solvent that was miscible with the alcohol and water. Additional challenges included obtaining the desired product without starting material or side reaction contamination. The addition of cysteine followed by careful control of the pH of the eluent appears to have remedied this problem; however, HPLC analysis of the products should settle the issue.
In a parallel exploratory study (see Scheme 2), the acid catalyzed synthesis of 6-O-methoxyguanine (compound 6) was discovered. Although the original target of this reaction was compound 2, conversion to 6 demonstrates a new method of activating 6-chloropurines to substitution.
References
- Belanch, Michael. Retrospective Study of the Correlation between the DNA Repair Protein alkyltransferase and Survival of Brain Tumor Patients Treated with Carmustine. Cancer Res., 56: 783, 1996.
- Robins, M. J.; Uznanski, B. Nucleic-Acid Related-Compounds. 33. Conversions of Adenosine and Guanosine to 2,6-dichloro, 2-amino-6-chloro, and derived purine nucleosides. Can. J. Chem., 59: 2601, 1981.