Collin Driscoll and Dr. Barry M. Willardson, Chemistry and Biochemistry
Introduction:
Cells use heterotrimeric G-proteins (Gabg) to transduce extracellular signals into intracellular messages. A hormone or other signal molecule binds to a seven trans-membrane receptor on the cell’s surface causing the receptor to change shape. On the inside of the cell this conformational change causes Gabg to split into a Ga subunit and a Gbg subunit complex. Both Ga and Gbg activate effector enzymes that determine the cellular response to the signal molecule. Phosducin (Pd) is a G-protein regulator that works by binding to the Gbg subunit complex and blocking Gbg function in three ways: 1) It blocks reassociation with Ga, a step necessary for reactivation of Gabg by receptors, 2) It blocks Gbg activation of effector enzymes, and 3) It dissociates Gbg from its functional place on the cell membrane. Pd has two domains which bind to Gbg: an N-terminus domain (Pd-N) which accounts for 2/3 of binding and a C-terminus domain (Pd-C) which accounts for 1/3. X-ray crystal studies of the Pd:Gbg complex have shown that Pd-N binds to the same surface as Ga, and that Pd-C covers the area which allows Gbg to bind back to the plasma membrane (1). This suggests that Pd-N is responsible for competing with Ga for Gbg binding (2) and that Pd-C is responsible for dissociating the Pd:Gbg from the membrane. However, there has been some data which suggest that the Pd-C, not Pd-N, competes with Ga for binding of Gbg (3). My research was designed to test whether the N-terminus or the C-terminus domain competes with Ga for binding of Gbg.
Procedure:
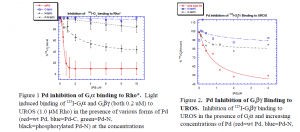
I used a G-protein system (receptor = rhodopsin (Rho) G-protein = Gt) found in retinal rods in my research. I performed an assay that tested the ability of the Pd-C and Pd-N to inhibit 125I-Gta binding to light activated Rho (Rho*). In the dark Gtbg, Rho containing membranes (UROS) and 125I-Gta are added to increasing concentrations of Pd. The samples are split into light and dark tubes. The dark samples are kept unexposed to light. The light samples are exposed to intense light for one minute and then both samples are centrifuged to pellet the membranes. Fifty mL aliquots are taken before and after centrifugation and analyzed for 125I-Gt in a g-counter. The difference in counts between light and dark gives the binding of 125I-Gt to Rho* and can be used to quantify how Pd inhibits 125I-Gt binding to Rho*(Figure 1).
I also performed a similar assay measuring dark binding of 125I-Gtbg to UROS in increasing concentrations of Pd. Maintaining all samples in the dark eliminated the presence Rho*, and therefore this assay measures only association of Gtbg to the lipid membrane (Figure 2).
Results and Discussion:
Figure 1 shows data from Pd-N, Pd-C, wild type Pd (which should inhibit maximally), and phosphorylated Pd-N (which should not inhibit at all). Since Gta must associate with Gtbg to bind Rho*, inhibition of binding indicates that Pd is blocking this association. Figure 1 indicates that the Pd-C does not significantly inhibit 125I-Gt from binding to Rho*. Therefore, the Pd-C does not compete with Gta for binding to Gtbg. The Pd-N data show inhibition to 42% with a K1/2 of 1.9 μM. Therefore, Pd-N does compete with Gta for binding to Gtbg. This is in agreement with the x-ray 83 0 20 40 60 80 100 0 2 4 6 8 10 Inhibition of 125I-G t binding to Rho* wtPd C-term N-term P-N term %125I-G t bound [Pd] mM Figure 2. Pd Inhibition of Gtbg Binding to UROS. Inhibition of 125I-Gtbg binding to UROS in the presence of Gta and increasing concentrations of Pd (red=wt Pd, blue=Pd-N, Figure 1 Pd inhibition of Gta binding to Rho*. Light induced binding of 125I-Gta and Gtbg (both 0.2 uM) to UROS (1.0 mM Rho) in the presence of various forms of Pd (red=wt Pd, blue=Pd-C, green=Pd-N, black=phosphorylated Pd-N) at the concentrations 40 50 60 70 80 90 100 110 0 1 2 3 4 Pd Inhibition of 125I-G t bg Binding to UROS wild type Pd N-term C-term % 125I-G tbg bound [Pd] mM crystal structure of Pd. The data also show that wild type Pd, which inhibits to 10% and has a K1/2 of 0.5 μM, is a more potent inhibitor than just the Pd-N. This suggests that in wild type Pd, Pd-N and Pd-C act synergistically to bind to Gtbg. Indeed, there are published data that give the Kd of wild type Pd binding to Gtbg at 100 uM (4). From this Kd, and knowing that Pd-N accounts for 2/3 of Pd’s binding to Gtbg and Pd-C accounts for 1/3, it is possible to estimate the Kd’s for Pd-C (4.6 mM) and Pd-N (21.1 mM).
The data from Figure 2 indicate that there is very little difference between Pd-N and Pd-C in preventing Gtbg from binding to UROS. The model based on the x-ray crystal structures of Pd:Gtbg would have predicted Pd-C to be a much more potent inhibitor than the Pd-N term. However, this apparent inconsistency can be explained by Pd-C’s low binding affinity for Gtbg. In the presence of the large amount of UROS in the assay (30 mM, with a Gtbg binding Kd of 2.0 mM) Pd-C (2.0 mM in this assay with a Gtbg binding Kd estimated at 4.6 mM) is out competed, and Pd-C’s binding to Gtbg is insignificant.
Conclusion:
These data support the hypothesis that: 1) Pd-N, not Pd-C, competes with Gta for binding of Gtbg, 2) Pd-C has a very low binding affinity for Gtbg, and 3) Pd-C and Pd-N act synergistically in binding of Gtbg. To finalize these findings, one final assay needs to be done; Pd termini competing with Gta for binding to Gtbg in the absence of UROS.
- Gaudet ,R.; Bohm, A.; Sigler, P.B. Cell. 1996, 87, 577-588
- Xu, J.; Wu, D.; Slepak, V.; Simon, M. Proc. Natl. Acad. Sci. 1995, 92, 2086-2090
- Bluml, K.; Schnepp,W.; Schroder, S.; Beyermann, M.; Macias, M.; Oschkinat, H.;Lohse, M.J. The EMBO J. 1997 16(16), 4908-4915.
- Yoshida, A., Willardson, B; Wilkins J; Jensen G; Thornton, B; Bitensky, M. (1994). J. Biol. Chem. 269, 24050-24057