Justin Gregory Peacock and Dr. Matthew R. Linford, Chemistry and Biochemistry
Introduction and Methods
Self-assembled monolayers (SAM’s) on gold have been extensively studied during the past 20 years because of their use in biochemical surface recognition, as nanotechnological structures, as nano-chemical wires and circuits in electronics, as a means of creating patterned surfaces, as sensors to monitor various chemical levels, and to attach proteins and other biomolecules to surfaces.1 In 1994 Offord et al.2 prepared series of mixed monolayers on gold using different proportions of t-butylthiol and 1-octadecanethiol. When monolayers were prepared from ethanolic solutions, anomalous surface properties were observed at intermediate t-butylthiol solution concentrations. They attributed the higher than expected thicknesses and lower than expected water contact angles to 1-octadecanethiol molecules physisorbed into vacancies in the monolayer created by t-butylthiol. Here we reconfirm the water contact angle and ellipsometric measurements that were previously performed and additionally use X-ray photoelectron spectroscopy (XPS) to establish the anomalous behavior of these mixed monolayers on gold.
Silicon wafers were cut into ~1 cm by ~3 cm shards using a diamond-tipped scribe. They were numbered from 1-22 using this pen and then cleaned in piranha solution (50:50 H2SO4 (conc.): H2O2 (conc.) (v/v)) at 110°C for ca. 15 minutes, and rinsed with distilled water. The wafers were coated in the BYU Physics Department’s thin film metals evaporator with about 200Å of Chromium followed by about 2000Å of Gold. Following metal deposition, but prior to surface modification, the optical constants of the gold were measured by variable angle spectroscopic ellipsometry at 45° to 75° in 10° increments. Two wafers each were immersed into solutions of tbutylthiol (t-C4SH) and 1-octadecanethiol (C18SH) (total thiol concentration was 1.0 mM) in ethanol with Rsoln = [t-C4SH]/[C18SH] values of 0, 1, 5, 7, 10, 25, 100, 500, 1000, 2500, and infinity.
Results and Conclusions
In order to fit the data to a thermodynamic model the following expression was used:2,3
PSAM = PSAM1 + (PSAM2 -PSAM1)*(1/(A*Rsoln + 1)) (1)
where PSAM1 is the property of a t-butylthiol monolayer on gold, PSAM2 is the property of a 1- octadecanethiol monolayer, and PSAM is the monolayer property corresponding to a particular ratio (Rsoln) of the concentrations of t-butylthiol and 1-octadecanethiol. “In a thermodynamic equilibrium model, A = exp(-á/RT) where á is the difference in free energies of adsorption of the two components”.2,3
The data for the advancing water contact angles (èa) versus the Rsoln follow the sigmoidal curve given in Equation (2):
èa = 56.25 + (111.125-56.25)*(1/((0.0020053*Rsoln)+1)) (2)
with an R-value=0.999, where the anomalous data points of Rsoln=5, 7, 10, 25 and 100 were not included in the fit. The plot of the monolayer thickness (Å) versus Rsoln, follows the sigmoidal curve in equation (3):
Thickness (Å) = 13.319 + (36.5-13.319)*(1/((0.00091683*Rsoln) + 1)) (3)
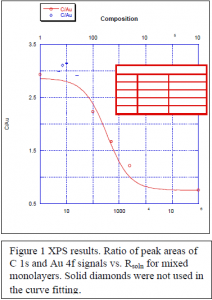
with an R-value=0.986. In contrast to the contact angle plot, the anomalous data points in the thickness plot are observed to lie above the fit. Figure 1 shows the ratio of peak areas for the C 1s and Au 4f XPS peaks. The data again follow a sigmoidal curve, as given by equation (4):
C1s/Au4f = 0.7565112 + (2.8649 – 0.7565112)*(1/((0.0029572*Rsoln) + 1)) (4)
with an R-value= 0.98733. These data again confirm the hypothesis that excess material from intercalated 1-octadecanethiol molecules is present in the monolayers. Once again, the fit to the data in Figure 1 was performed by omitting anomalous data points (filled diamonds).
We have reconfirmed the trends that were observed by Offord et al. in their study of mixed monolayers of tbutylthiol and 1-octadecanethiol using contact angle goniometry and spectroscopic ellipsometry, and applied a new technique (XPS) to further show these effects. We propose that the resulting surface should have free sulfhydryl groups that will be capable of reacting with different chemical, biochemical, and polymeric reagents. Thus we see the preparation of mixed monolayers with intermediate concentrations of t-butylthiol and 1-octadecanethiol as a straightforward method for preparing functionalized (sulfhydrylterminated) monolayer surfaces.
References
- Mrksich, M. Curr.Op.Chem.Biol. 2002, 6, 794-797.
- Offord, D. A.; John, C. M.; Linford, M. R.; Griffin, J. H. Langmuir 1994, 10, 883-889.
- Folkers, J. P.; Laibinis, P. E.; Whitesides, G. M. Langmuir 1992, 8, 1330-1341.