Karen Merrell and Dr. Steven W. Graves, Chemistry and Biochemistry
Na,K-ATPase is a multi-subunit protein that uses ATP to pump Na+ ions out of the cell and K+ ions into the cell. Multiple isoforms of each subunit of Na,K-ATPase have been identified, but the á subunit of Na,K-ATPase plays the most important functional role and has been the most widely studied. Within the lens, Na,K-ATPase is most concentrated in the outer epithelial layer. To study changes in Na,K-ATPase levels I cultured bovine lens epithelial cells (BLEC), and incubated them with various hormones. The abundance of the á3 isoform of Na,K-ATPase was determined by Western blot protein gel electrophoresis using specific monoclonal antibodies to measure the quantity of the á3 isoform.
Primary cell culture is a very difficult thing to do and the biggest challenge I encountered was getting the BLEC cells to grow. I collected fresh bovine eyes from the Deseret Meat packing plant and put the epithelial layer into culture media within three hours so the cells would still be alive. Because the cells came from a source that was non-sterile I had a hard time avoiding contamination in the cell culture media. I could not get any cells to grow for a few months. I tried changing the cell media more often, getting the bovine eyes earlier to make sure the cells were alive, and different dissection techniques, but no cells would grow. I bought new antibiotic to put in the media and cells began to divide and grow.
Because there is so little Na,K-ATPase present in each cell, I had to use a lot of cells to be able to detect changes in the expression of this protein. Growing enough cells took from four to six weeks to go from dissection to experiment. When there were a sufficient number of tissueculture plates with confluent cells I began incubations with physiological levels of various hormones. The hormones used were DHEAS, estrogen, progesterone, and testosterone. Three 10- inch tissue culture plates were used for each hormone and then harvested after 24 hours. The three plates of cells were pooled into one sample, and then were frozen to lyse the cells. the cell membranes were centrifuged down, and the cytosolic portion of the sample was removed. The cell membranes then had to be homogenized to release any Na,K-ATPase. Homogenization didn’t work well using either a tissue-tearer or a teflon-glass tissue grinder, but did work successfully with a sonicator.
The total protein was quantified in each sample, and equal amounts of protein for each sample were run on a polyacrylamide gel to separate all of the proteins by size. After the proteins were separated, they were transferred onto a nitrocellulose membrane. This membrane was incubated with a solution containing nonfat dry milk to block any non-specific protein binding. The membrane was then incubated with an antibody specific to the á3 isoform of Na,K-ATPase, and this was then incubated with a secondary antibody that bound the primary antibody. The secondary antibody emits light when activated, and this light was detected by exposing the nitrocellulose membrane to photo-paper in a darkroom and then developing the paper.
Using this method one would ideally see clearly defined bands on the photo paper only where there was á3 Na,K-ATPase present. Unfortunately, this is often not the case. With such a long procedure, it is easy to make mistakes that will destroy your experiment, or obscure your results.
For example, the membrane fraction of each sample produced a very faint signal when compared to that of the cytosolic fraction, though the amounts of total protein added was the same. This suggests that there is a higher percent of other proteins in the membrane of BLEC cells, and that more sample needed to be loaded onto the gel. The main problem with this is that I would need to use more total cells to generate the amount of sample needed for an adequate signal, and large quantities of cells take a long time to grow. Another problem encountered in these experiments was getting clear, readable signal from the gels. Air bubbles between the polyacrylamide gels and the nitrocellulose membrane during transfer prevent protein transfer in those areas, running the gel for too little time doesn’t allow the protein bands to separate enough, allowing the apparatus to get too hot can break the glass plates used to cast and run the gels, allowing too little time during transfer can produce poor signal, using too little antibody reduces signal too much, but using too much antibody increases non-specific binding and is costly. There were many errors made as I learned the necessary skills to get useable data from these experiments.
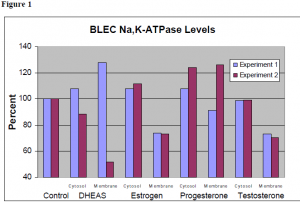
The data from the two experiments that resulted in clear data are shown in Figure 1. Each of the fractions are shown normalized to a similar (cytosol or membrane) fraction of a control. The data for the incubations with DHEAS and progesterone were not very consistent, so no preliminary conclusions can be drawn on the effect of these hormones to á3 Na,K-ATPase levels in BLEC. These data suggest that incubation with estrogen slightly increases the á3 Na,KATPase level in the cytosol and decreases levels in the membrane. Incubation with testosterone does not change levels in the cytosol, but decreases levels in the membrane.
There is much further work that needs to be done to draw any firm conclusions about the effects of these hormones on the expression of Na,K-ATPase in BLEC. These same experiments need to be performed a few more times in order to get consistent data. The effect of a combination of these hormones could also be studied. I studied only the distribution of the á3 isoform of Na,KATPase, but there are two other á subunits whose levels could also be studied. I have trained two undergraduate students, and they are currently continuing these studies.