Sean P. Brady and Dr. Paul B. Savage
With help from fellow lab members, this project involved the synthesis of several antigen linkers in order to aid the understanding of histamine secretion of basophil cells. It is hypothesized that the bi-valent linkers (such as the ones outlined below) do not stimulate basophil cells, but that the tri-valent antigens (not shown in this report) do cause granulation of cells.
The proposed compound was synthesized as outlined, beginning with serinol (figure 1) followed by the coupling of the variant linker (ie. figure 2). First, the hexa-linker was synthesized by coupling Sanger’s Reagent and the hexa-ethylene glycol components together to yield the compound shown below.
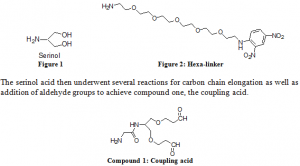
These preparatory reactions were relatively easy to complete and gave very high yields. The next and final steps, however, which involve the actual coupling of the linker and acid, did not go as smoothly. Several attempts were made to ensure purity of the two compounds and that reaction conditions were met to the set specifications. After much effort, product was made and purified for classification. More than one gram of the hexa-linker, a considerable amount for testing, was obtained. This final reaction included the attachment of two linker groups to either of the aldehyde ends of the coupling acid to form the final product. Difficulties in this last step could have included purity issues of the acid since it is fairly reactive with itself, and if it is let to sit by itself over time, it can degrade and form by-product.
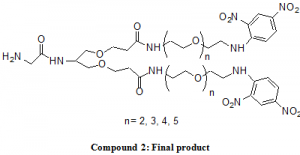
The hexa-linker final compound is ready to be sent to the collaborative research facility in Flagstaff, Arizona, where it will be tested for basophil stimulation. Linkers of other lengths are also to be tested. The tests will involve injecting the compound into live rats that will be monitored for any increases of basophil concentration in the blood. The current hypothesis is that the bi-valent antigen linkers will not stimulate any activity of the basophil cells.
After completion of this project, another has been proposed by the Arizona staff to continue synthesis of these linkers by adding another fluorophore group to the main serinol acid. This project is now awaiting funding and will commence upon approval.
With further data and experimentation of these linkers, publication of these compounds and their findings could be entered into JACS (Journal of American Chemistry Society), Nature, or other prominent chemistry periodicals.
