Bryce Harbertson and Dr. Heidi Vollmer-Snarr, Chemistry and Biochemistry
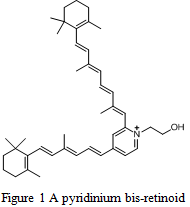
Photodynamic therapy involves the use of light-sensitive compounds that, upon irradiation, become lethal to cells. These light-sensitive compounds are referred to as photosensitizers. A class of compounds known as pyridinium bis-retinoids are being investigated as potential photosensitizers, and therefore, chemotherapeutic drugs (Figure 1). One of the primary concerns in these investigations is determining the optimal conditions under which to evaluate these drugs. The optimization of these parameters will allow us to compare the effectiveness of our drugs with others already in existence, and will also serve as a basis for further investigations in animal trials and later on in clinical trials.
The variables studied during this project included light intensity, time of light exposure, and length of drug incubation. We also had planned to test different types of light sources but in the end were unable to do so because of time constraints. Numerous experiments were run to obtain reproducible results. Furthermore, we tested our pyridinium bis-retinoid compounds on different types of cells. Different types of cells have different properties and therefore necessitated more time to test the aforementioned parameters.
We initially set out to optimize these parameters in kidney cancer cell assays; however, this proved to be a significant burden. We frequently found that the kidney cancer cells we were using had become contaminated with bacteria. This prevented us from running cell assays as we did not have an adequate number of cells. Even if we had had sufficient cells, running the assays would have been futile as we would not have been able to determine if cell death was due to a parameter change or contamination. We later discovered that the source of contamination was the liquid media we used to help the cells grow. Resolving this issue was simple, as we now filter our media before using it with any cell type and supplement the media with antibiotics. The cells we now grow have not had any problems with contamination.
Although we resolved the contamination issue relatively easily, we had already decided to test our drugs on different cell types. We therefore conducted these experiments utilizing breast cancer cells (MDA-MB-435) and leukemia (HL-60) cells.
Our experiments have found that the optimal irradiation time period is similar for both the breast cancer and leukemia cell lines. The irradiation time period simply refers to the length of time that the cancer cells are exposed to light. In our case, we utilized a fiber optic light source and a dichoric blue filter which permitted the passage of 400-500 nm light (blue light). With this filter, the irradiation times that yielded the best results were 5 and 10 minutes (Figure 2). Longer irradiation times, such as 45 minutes, resulted in death among cells with no drug, meaning that death was caused by light exposure alone and was not due to the effects of the drug.
In addition to testing the length of irradiation, we also set out to determine the optimal length of time between drug addition and irradiation. This parameter was designated as the drug incubation period and has been one of the most interesting to investigate. Many cells have the ability to pump out undesirable compounds. We therefore tested two incubation periods – 6 hours and 24 hours – and found that 24 hours was an optimal time for leukemia cells while 6 hours was preferential for the breast cancer cells (Figure 2). No significant death was observed in the breast cancer cell line when an incubation period of 24 hours was used, nor was there significant death in the leukemia cells when using an incubation period of 6 hours. We have not found an exact explanation for why this occurs, but believe it may be the different pumping or excretory mechanisms inherent in each cell type.
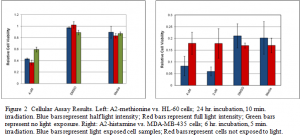
The last parameter we were able to investigate was drug concentration. A very high drug concentration may cause cell death in cells that are not directly exposed to light. Although the drugs are not inherently lethal to cells, a small percentage of the drug may be exposed to residual light. Therefore, it is important to use the lowest concentration possible to minimize unwanted death in cells not exposed directly to light. We found the most effective drug concentration to 4 μM, as higher concentrations caused death without direct light exposure and lower concentrations did not cause significant death after light exposure. While this is a low concentration, we hope future experiments will show that even lower concentrations can cause death in cancer cells.
Our project concluded that short irradiation times and relatively low drug concentrations were preferable conditions for both cell lines tested. Additionally, we found that the optimal incubation period varies depending on the cell type, and believe this may be related to how the cell gets rid of unwanted drugs and other compounds. Further experiments will be performed to reproduce these results, test other pyridinium bis-retinoids, and test these compounds on other cancer cells to elucidate their full range of effectiveness.
