D. Karl Bedke
Asymmetric glycolate alkylation plays a key role in the synthesis of complex biologically active compounds. Such asymmetric transformations have largely to date been accomplished by employing chiral auxiliaries which have shown remarkable success in asymmetric synthesis. However, auxiliaries must also be used in stoichiometric amounts and can become quite expensive. A catalytic approach to asymmetric glycolate alkylations presents an attractive option to the use of auxiliaries. Andrus2,3 et al. have recently developed a methodology utilizing phase-transfer catalyst 1 for alkylation of a novel alkoxyacetophenone substrate 2 that can be readily oxidized to its glycolic ester derivative to provide access to enantioenriched glycolic acid derivatives. To illustrate the utility of this methodology, two biologically active targets were selected and synthesized; their total syntheses are reported herein.
The peroxisom proliferators-activated receptor (PPAR) agonist (-) ragaglitazar functions as a dual agonist in the attenuation of hyperglycemia and hyperlipidemia associated with type 2 diabetes. This compounds was colicensed by Novo Nordisk and completed phase II clinical trials.4 Previous syntheses of this compound employs an expensive Horner-Emmons reagent, and relies on a kinetic racemic resolution by a hydrolase wherein one-half of their material is lost. Furthermore, the carbon framework of ragaglitazar is prevalent in many related therapeutic compounds.5
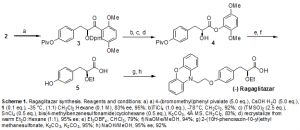
The synthesis of ragaglitazar (Scheme 1) was commenced by treating substrate 2 under optimized phase-transfer catalyzed conditions, using solid cesium hydroxide monohydrate as the base, a pivalate protected benzyl bromide and the Park and Jew triflurobenzyl-hydrocinchonidinium bromide salt 1. We were delighted to find that this reaction produced 3 in good yield with good selectivity. Subsequent removal of the diphenylmethyl (DPM) group under Lewis acidic conditions followed by a Baeyer-Villager like oxidation yielded the - hydroxy aryl ester 4. At this point, we were again pleased to find that this ester could be recrystalized from warm ether to give essentially enantiomerically pure products (~95% ee). The free hydroxyl was then alkylated using triethyloxonium tetrafluoroborate, and then transesterification under catalytic basic conditions produced 5. A mesylated phenoxazine alcohol reacted with 5 to yield the methyl ester of 6, which was obtained by treatment with sodium hydroxide in methanol. The overall synthesis proceeds with 47% overall yield (41% from commercially available reagents) and is eight linear steps from the alkoxyacetophenone substrate 2, including a recrystalization.
The farnesyl-protein transferase (FPTase) inhibitor kurasoin A was isolated from the culture of Paecilomyces sp. FO-3684 in 19966. It operates by mutating the activity of FPTase, which in turn inhibits farnesylation, and eventually stymies the proliferation of the cell. Farnesyltransferase inhibitors provide an attractive approach to the development of cancer chemotherapeutics because they are selective to oncogenic cells. Due to the selectivity of this compound, we embarked on a synthesis of this compound that would provide flexibility to future derivation.
The synthesis of kurasoin A (Scheme 2) is analogous to ragaglitazar through to the generation of 4. Protection of 4 with triethylsilyl chloride, followed by Wienreb amide formation with trimethyl aluminum and N,O-dimethylhydroxylamine afforded 6. Benzyl Grignard was then added into 6 and removal of TES and pivalate with TBAF and LiOH/H2O2 respectively yielded kurasoin A.
In conclusion, the total syntheses of the PPAR agonist ragaglitazar and FPTase inhibitor kurasoin A are complete, and illustrate the utility of asymmetric glycolate alkylations under phase-transfer catalyzed conditions. These results suggest that this methodology has potential to be assimilated into total synthesis strategies for larger molecules.
Acknowledgements: Special thanks to Dr. Merrit B. Andrus, Dr. Erik J. Hicken, Jeffery C. Stephens and the Office of Research and Creative Activities for financial support.
References
- Dolling, U. H.; Davis, P.; Grabowski, E. J. J. Efficient Catalytic Asymmetric Alkylations. 1. Enantioselective Synthesis of (+)-Indacrinone via Chiral Phase-Transfer Catalysis. J. Am. Chem. Soc. 1984, 106, 446–447.
- Andrus, M. B.; Hicken, E. J.; Stephens, J. C. Phase-Transfer Catalyzed Asymmetric Glycolate Alkylation. Org. Lett. 2004, 6, 2289–2292.
- Andrus, M. B.; Hicken, E. J.; Stephens, J. C.; Bedke, D. K. Asymmetric Phase-Transfer Catalyzed Glycolate Alkylation, Investigation of the Scope, and Application to the Synthesis of (-)-Ragaglitazar. J. Org. Chem. 2005, ASAP.
- Henke, B. R. Peroxisome Proliferator-Activated Receptor Dual Agonists for the Treatment of Type 2 Diabetes. J. Med. Chem. 2004, 47, 4118–4127.
- Wilson, T. M.; Brown, P. J.; Sternbach, D. D.; Henke, B. R. The PPARs: from orphan receptors to drug discovery. J. Med. Chem. 2000, 46, 1306–1317.
- Uchida, R.; Shiomi, K.; Inokoshi, J.; Masuma, R.; Kawakubo, T.; Tanaka, H.; Iwai, Y.; Omura, A. Kurasoins A and B, New Protein Farnesyltrasferase Inhibitors Produced by Paecilomyces sp. FO-3684. J. Antibio. 1996, 49, 932–934.
