Adam Bevan and Dr. Scott Steffensen, Psychology
In today’s world addiction is pandemic. So many people are slaves to these controlling chemicals, from alcohol and street drugs to the more mild substances of coffee or over-thecounter medications. All these substances tend to influence brain chemistry, and the aim of our ongoing project is to uncover the effect of alcohol, cocaine and their anologues on midbrain circuitry. We believe that gamma-aminobutyric acid (GABA) neurons are primarily involved in addiction to these substances.
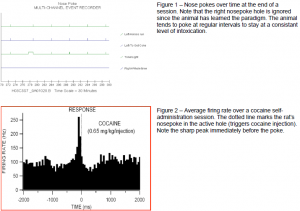
My initial project was on the discharging, or coupled firing, of ventral tegmental area (VTA) GABA neurons during alcohol self-administration. Many experiments in the Steffensen lab have been done under anaesthesia on a stereotaxic setup. These are very well controlled experiements, but we wanted to verify that we could see the same effects in awake animals. These proved to be very difficult tasks due to the nature of the surgery, length of recovery and time to ethanol dependency. For our surgeries, male Wistar rats were anesthetized by continuous inhalation of 1- 3% Halothane gas and a permenant urethane catheter was threaded into the femoral vein. The animals were then transferred to a stereotaxic setup (still under general anaesthesia) and a recording electrode was placed in the VTA and a stimulating electrode was placed in the internal capsule (IC). Both electrodes were permanently attached to the skull. The animals were allowed to recover for 3-7 days before behavioral experiments commenced. These involved placing the animals in chambers equipt with two nosepoke holes and a drug-injection system. One hole (left) was active, and would trigger the system to inject a drug into the animal via the i.v. catheter. The other hole (right) was inactive, but the system still monitored nosepoke activity in both so we could monitor exploratory vs. drug-seeking behavior (Figure 1). During each session, VTA GABA neuron firing rates were monitored and aligned with nosepoke activity (Figure 2). Periodically during certain sessions, the IC was stimulated and the resultant VTA firing was observed.
We found that it took so long for the animals to become physiologically or psychologically dependent on alcohol that the electrodes were failing (likely due to gliosis and/or swelling) so we couldn’t obtain enough electrophysiological data. We tried this same technique with cocaine instead of alcohol, and the rats only needed a few hours in the chambers to learn the key behavior. This means that reliable data was easier to obtain. We therefore decided to continue with cocaine self-administration instead of our original aim to stay with alcohol.
Naïve rats had to be carefully monitored to ensure that they did not over-dose on cocaine. At later points in the paradigm, however, the animals tended to poke at regular intervals, likely to sustain a preferred level of intoxication (Figure 1). Our data show that VTA GABA neuron firing rate increased 50-100ms prior to poking for cocaine. It is possible, though not etirely clear from the data, that the same neurons may even decrease firing rate immediately following the nosepoke (Figure 2). The prevailing dogma in addiction research is that animals seek cocaine because of its direct effect on dopaminergic midbrain circuits. Although our data is not conclusive, we tend to to believe that GABA circuits are intimately involved in mediating drugseeking behavior because of the immediacy of the changes in GABA firing rates.
Another important finding was the verification of previous data obtained from anaesthetized animals. Rapid IC stimulation unleashes very strong, highly coordinated VTA GABA neuron firing in anaesthetized animals. We successfully reproduced this effect in our awake animals at varying levels of intoxication. We even found resonant activity that lasted up to two or three times as long compared to when the animal was anaesthetized. This discharging activity, like in the anaesthetized animals, diminishes with increasing levels of cocaine intoxication. The occurance of these phenomena in both awake and anaesthetized subjects enables us to better correlate our previous findings in acute stereotaxic preparations with those from freely-moving experiments in behavioral chambers.
Dr. Steffensen’s laboratory is still working with these models to further understand how GABA neurons are involved in addiction and substance abuse, and we are still far from understanding it completely. However, the data generated during this project give us a slightly better idea of how everything works together. I would like to thank Dr. Steffensen for his quality mentorship during my years in his lab. I would also like to thank BYU, ORCA and NIDA for their financial support. I am confident that these small steps will eventually lead us to a more complete understanding of addiction and provide viable therapies to aide those caught in the trap of substance-abuse.