Thomas Wall and Dr. Aaron Hawkins, Electrical Engineering
Introduction
Rubidium vapor has certain physical properties that allow it to be optically probed in order to produce many interesting quantum effects; however, due to its reactive nature rubidium can be very difficult to encapsulate. Historically bulk rubidium vapor cells have been used, which rely on high temperature glass forming and sealing. The potential that rubidium vapor has in real-world application can be greatly increased by creating a smaller, more versatile vapor cell that does not require high temperatures to be created. We are working to create a rubidium vapor cell that can be integrated with a chip-scale device in order to open the door to many important applications such as optical buffers, tunable diode lasers, magnetometers, atomic clocks, slow and stopped light, and many other quantum communication elements.
Successful introduction and sealing of rubidium in our chip-scale device, called an ARROW waveguide, has several requirements:
(1) We must create an air tight seal between Silicon and Copper.
(2) No materials can be used that chemically react with rubidium.
(3) The seal must be long lasting and robust.
(4) The vapor cell must be able to withstand fabrication temperatures of up to 250°C.
(5) The vapor cell must be small enough to integrate with our ARROW waveguide devices.
Methods
The quantum optics research team has developed a unit called a “minicell” in order to identify the ideal method for creating a hermetic seal between silicon and copper, using materials that are non-reactive with rubidium. The mini cell uses a glass vial to represent the silicon of the chip. The rubidium is sealed inside the “minicell”, and laser light is passed through the glass vial in order to perform atomic spectroscopy on the rubidium vapor inside the cell.
The atomic density of the rubidium vapor is calculated using the spectroscopy performed and the rubidium is then classified over time. This testing reveals whether or not the rubidium inside the seal is sealed tight and remains unreactive or whether it is reacting with a material over time.
Several different methods of creating a seal between the glass vial and copper tube were investigated. Three of the most important techniques and their results will be described here. The simplest method was attaching the copper tube and glass vial using solely epoxy. The other two methods involved the investigation of using various metals and soldering techniques to attach the copper and glass. This was done by depositing nickel onto the surface of the glass vial using a combination of electron-beam evaporation and electroplating in the clean room facilities at BYU. The copper reservoirs are then be soldered at the lowest temperature possible using some type of solder. Two different solders were investigated at this point: Indium and Lead/Tin (Pb/Sn). Both of these solders react with the rubidium and must be covered with a layer of copper in order to prevent exposure to the Rubidium. In order to accomplish this copper is selectively electroplated over the two different solders. This creates an airtight seal that exposes the rubidium vapor to only glass and copper.
Results
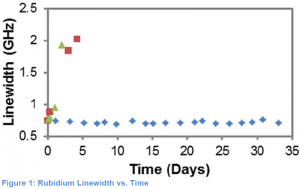
Many “minicells” were created using these different sealing methods, stored at various temperatures and then were tested on our atomic spectroscopy set up at BYU in order to determine their lifetime. The results from the atomic spectroscopy allow us to monitor what is called the full width at half maximum linewidth of the rubidium inside the “minicells”. As rubidium chemically reacts the linewidth of the rubidium will increase. By monitoring the linewidth of the Rubidium we could tell determine whether or not the rubidium inside the “minicell” was chemically reacting or not. Figure 1 shows the linewidth of the three different sealing techniques over time. The linewidth of the this indium solder cell(blue diamonds) remained constant while the linewidth of the epoxy cell(red squares) and Pb/Sn solder cell(green triangles) increased dramatically in only a few days.
Discussion
The rubidium vapor cells created with indium solder remained active for over 30 days and therefore we conclude that this method can be used to create vapor cells with extremely long lifetimes. The successful demonstration of a long-lasting “minicell” means that we are now ready to translate this sealing method to our ARROW devices and demonstrate some of the quantum effects listed in the above introduction.
