Adam Calvert and Dr. Merritt Andrus, Chemistry and Biochemistry
Natural compounds can be found throughout the world in an essentially limitless variety. By discovering compounds and investigating their properties, we can unlock the mysteries to cure rare diseases and alleviate common ailments. There are however many challenges with using natural compounds in such ways. The first problem is that most biological molecules are found in very small amounts. This not only poses as a problem with identifying their unique properties, but also stands in the way of their pharmaceutical use. A key solution to this challenge is organic total synthesis.
Organic total synthesis is the process of converting common (and often inexpensive) starting materials into the rare natural products. This is done in a series of reactions to build the desired compound. The reactions can include those found in conventional organic chemistry procedures, literature publications, and novel mechanisms designed by the researcher.
As total synthesis is explored further, a second challenge arises in the asymmetric configuration of nearly all natural products. A chemical compound is considered asymmetric, or chiral, when its mirror image is not super-imposable on itself. Many biological compounds can be seen as a lock and key mechanism; if a mirror of the key were created, it would not fit into the original lock, and likely have an adverse physiological effect. Thus, when synthesizing a natural compound special care must be taken to insure that the correct stereochemistry, asymmetric form, is created.
Dr. Andrus has developed a new methodology to stereoselectively add groups to a molecule in the synthesis of natural products. The novel technique uses an asymmetric phase-transfer catalyst (PTC). A conventional PTC is used to react a hydrophobic compound with a hydrophilic one. It is commonly used in alkylation reactions, in which an alkyl group (carbon based group) is added to another molecule. When a conventional PTC is used, such as tetrabutyl ammonium bromide, the new alkyl group will be racemic, having equal amounts of both asymmetric forms. By using the asymmetric PTC, Dr. Andrus can successfully perform alkylation reactions resulting in high amounts of the preferred asymmetric form.¬1
With Dr. Andrus’ new technology, we began designing a total synthesis of the compound 12-HETE. 12-HETE was first discovered in 1974 by incubating human platelets in an acid. 2 This compound plays a role in several biological pathways including angiogenesis, cancer metastasis, atherogenesis, coronary thrombosis, type I diabetes, inflammation, psoriasis, and apoptosis. 3 Like other target products in Andrus’ lab, 12-HETE exists in nature in a specific asymmetric form (1).
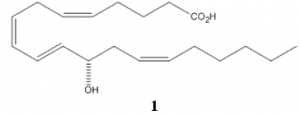
The key step of the synthesis of 12-HETE is the alkylation reaction in which the compound becomes asymmetric. As described above this reaction utilizes the new techniques designed by Dr. Andrus with an asymmetric PTC. In order to optimize the reaction, screenings were run for several different factors. We sought the reaction conditions with the greatest enantiomeric excess (% ee), which measures the amount of desired stereochemistry in excess of its mirror form. The variables screened include the catalyst, solvent, and temperature. Table 1 contains the results of the screenings.
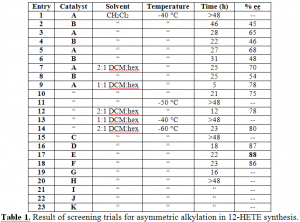
Twenty-three trials were run with 11 different catalyst, 3 different solvents, several temperatures, and many different reaction times. Screening entry 17 resulted in the greatest enantomeric excess at 88%. The optimal conditions found were catalyst E, -60°C, and 2:1 dichloromethane and hexanes as the solvent.
These results further validate Dr. Andrus’ technology as a viable method of obtaining the desired asymmetric form in total synthesis. The project is now prepared to move toward completion by using conventional organic chemistry procedures to finished the synthesis of 12-HETE. Once the synthesis is complete further investigations can be done into the unique properties and pharmaceutical potential of 12-HETE.
References
- Christiansen, M. A.; Butler, A. W.; Hill, A. R.; Andrus, M. B. Synlett 2009, 4, 653-657.
- Hamberg, M; Samuelsson, B. Proc. Nat. Acad. Sci. USA.1974, 71, 3400-3404.
- Han, X.; Corey, E. J. Org. Lett. 2000, 2, 2543-2544.
