Aaron Butler and Dr. Merritt Andrus, Department of Chemistry and Biochemistry
S-equol is a soy isoflavone metabolized from the isoflavone daidzein by intestinal microflora. Isoflavones are a class of nonsteroidal estrogens. A study conducted by DellaPenna indicates that populations with isoflavone-rich diets have fewer incidences of cardiovascular disease, osteoporosis, and some cancers including breast, prostate, and colon cancers (1). A study by Setchell et al suggests that the health benefits seen in populations whose diets are rich in isoflavones may be correlated with people’s ability to produce s-equol (2). Despite encouraging preliminary studies regarding s-equol, there has yet to be extensive studies done with s-equol in vivo. This is due to the difficulty to synthesize s-equol and its consequent high cost. In order to ascertain the specific health benefits s-equol may provide, further studies need to be done and a better synthesis of s-equol must be manufactured.
It is the goal of this project to develop an improved asymmetric synthesis of s-equol, and thereby obtain sufficient quantities for biological testing. The s-enantiomer is the focus of this project since intestinal bacteria produces s-equol exclusively in nature (3,4). The critical focus of this project will hinge on generating the stereocenter in a highly enantiopure fashion.
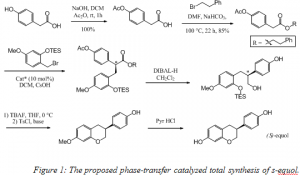
A new asymmetric total synthesis of the soy flavanoid (s)-equol is proposed (see figure 1), requiring only nine linear steps. The key model step, the creation of the stereogenic center, still only gives a 48% ee using the best conditions studied thus far. This will be improved by exploring various substrates, catalysts, bases, solvents, and temperatures. Once these two obstacles have been overcome, this phase-transfer-catalyzed total synthesis of s-equol should expand the amount of research able to be performed with s-equol. It is anticipated that this work will also help expand the scope of asymmetric phase-transfer-catalyzed alkylation particularly the use of cinchona-derived catalysts.
References
- DellaPenna, D. Science 1999, 285, 375–379.
- Setchell, K. D. R.; Brown, N. M.; Lydeking-Olsen, E. J. Nutr. 2002, 132, 3577–3584.
- Muthyala, R. S.; Ju, Y. H.; Sheng, S. B.; Williams, L. D. Bioorgan. Med. Chem. 2004, 12, 1559 1567.
- Setchell, K. D. R.; Clerici, C.; Lephart, E. D.; Cole, S. J. et al.; Am. J. Clin. Nutr. 2005, 81, 1072–1079.
