John T. Prince and Dr. Merritt B. Andrus, Chemistry and Biochemistry
The failure of chemotherapy in treating cancer is largely due to a phenomenon known as Multidrug Resistance (MDR). In MDR, cancer cells that have been exposed to a particular chemotherapeutic drug build up resistance not only to the drug originally administered, but to a host of other chemodrugs that may or may not be structurally related.1 P-glycoprotein (Pgp), a large, membrane bound transporter, is primarily responsible for MDR. Pgp acts as a pump to efflux cytotoxic drugs, keeping internal concentrations at less than cytotoxic levels. Although other factors contribute to MDR, it is generally thought that the most promising approach to MDR reversal at this time is the development of potent Pgp transport inhibitors. 2
Because Pgp is capable of binding and transporting a wide range of dissimilar compounds, 3 and since the fine structure and transport properties of Pgp have remained obscure, the design of effective inhibitors has been particularly difficult. Thus, an approach allowing for the rapid synthesis and screening of large numbers of potential inhibitors seems ideal. I have been synthesizing a 10 X 10 solution phase combinatorial library based on 4,5-dehydro-stipiamide that elaborates on the results achieved by our recently completed 6 X 7 library.4 When complete, this 10 X 10 library will test 100 unique compounds for potency while having required only 20 final couplings and 20 assays–a 5 fold increase in efficiency over a one-molecule-one-assay approach. The basic mechanics of the library are illustrated (Fig. 1) and described below. Vinyl iodide 1 and enyne 2 are synthesized as an alcohol and carboxylic acid respectively. Ten unique sulfides are then ether linked to 1 giving ten unique vinyl iodides. Ten unique amines are coupled to 2 giving ten unique alkynl amides.
These twenty compounds are then palladium coupled so as to create twenty pools of compounds. Each of ten pools contains a unique vinyl iodide coupled to the ten different alkynl amides. Each of the other ten pools contains a unique alkynl amide coupled to all ten different vinyl iodides. Pools are then tested for potency against adriamycin resistant breast cancer cells. Plotted in two dimensions, these results allow individual compounds to be identified that are predicted to have potent MDR reversal activity. Lead compounds will then go on to further testing.
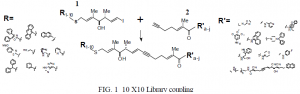
These twenty compounds are then palladium coupled so as to create twenty pools of compounds. Each of ten pools contains a unique vinyl iodide coupled to the ten different alkynl amides. Each of the other ten pools contains a unique alkynl amide coupled to all ten different vinyl iodides. Pools are then tested for potency against adriamycin resistant breast cancer cells. Plotted in two dimensions, these results allow individual compounds to be identified that are predicted to have potent MDR reversal activity. Lead compounds will then go on to further testing.
This past winter and summer I have completed the synthesis and characterization of all ten alkynl amides. The two couplings used and resulting alkynl amides are detailed below. (Fig. 2)
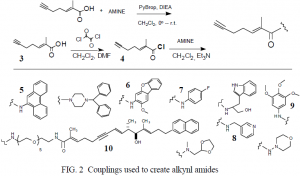
I initially selected amines based on lipophilicity, number and structure of rings, partition coefficients, polarity, and known structure-activity relationships. However, some amines conjugated to aromatic groups proved difficult to couple due to their decreased nucleophilicity. I bypassed this problem in three ways. First, by converting the alkynl acid 3 into an acid chloride 4 (a less stable compound with higher electrophilicity) I was able to couple some amines containing few or no electron withdrawing groups (5, 6, 7). Second, I replaced aminopyridine with 8 to disrupt the conjugation and allow coupling. Finally, amines were selected which contain an electron donating group in their ring system (6 and 9) to make the amine more nucleophilic.
I spent a considerable amount of time synthesizing the napthyl dimer 10. In previous work done by our group, this dimer (in its homodimer form) was shown to have very high potency. By including it in this library we hope to show the versatility of a solution phase combinatorial approach and to identify even more potent compounds. Thus, the eight reaction steps and considerable time necessary to synthesize the dimer seems justified.
In the future, I will be working towards the synthesis of the 10 vinyl iodides and in coupling the 20 pools of compounds. Additionally, I am coupling the alkynl amides to a napthyl vinyl iodide to begin work on a sectrometrically monitored selection experiement (SMOSE) with Pgp.
References
- Kartner, N.; Ling, V. Sci. Amer. 1989, 260, 44-51.
- Ford, J. M. Eur. J. Cancer. 1996, 32A, 991-1001.
- Klopman, G.; Leming, M.; Rama, A. Molecular Pharmacology. 1997, 52, 323-334.
- Andrus, M. B.; Turner T. J. Org. Chem. 2000, 65, 4973-4983.
- Much thanks to Timothy Turner for all his help and guidance.
