Nicole L. Hatch and Dr. Daniel L. Simmons, Chemistry and Biochemistry
Non-steroidal anti-inflammatory drugs (NSAIDS) are the basis for a multi-billion dollar pharmaceutical market. Their target, Cyclooxygenase (COX), might from an economic standpoint be considered the most important enzyme known to man.1 COX is the key enzyme in the transformation of arachidonic acid into prostaglandins, is the target of non-steroidal antiinflammatory drugs (NSAIDs). Prostaglandins are very important signaling molecules that are involved in many body functions. From physiological and pathological standpoints the most interesting functions are in arthritis, inflammatory pain, ovulation, and cancer. Two isoforms of COX are known to exist in mammals: COX-1 and COX-2. Although very similar in structure, each enzyme has unique functions. COX-1 prevents gastrointestinal bleeding while COX-2 causes pain and inflammation. Identifying a drug which is more selective for COX-2 than COX- 1 would give a drug without the current life threatening side effects of traditional NSAIDs. Since the second greatest market for NSAIDs is the treatment of osteoarthritis in canines, work was focused on this species.
To develop canine COX-2 selective drugs for the treatment of arthritis and pain in dogs, it is necessary to develop accurate models for evaluating COX-1 and COX-2 inhibition. The serum albumin in dog’s blood is well known to bind NSAIDs with much less efficiency then in humans, therefore dogs must be examined separately from other species.
In order to test the canine COX-1 and COX-2 selectivity of eleven NSAIDs, an established human whole blood assay protocol was modified for use as a canine whole blood assay using our model cell lines. 2 I chose to focus my report of results on aspirin, carprofen and diclofenac, though eleven NSAIDs were tested concurrently.
CCL-34, Madin Darby canine kidney cells (American Type Culture Collection) were grown and plated into 96 well-plates at 50,000 cells/ml. For the COX-2 assay they were then treated with media containing phorbol ester (TPA) at a concentration of 10 mg/ml. Dogs from the veterinary lab donated approximately 50 ml fresh blood for each experiment. Heparin was added to the blood to keep it from coagulating. Stock drug solutions of 22 mM concentration in DMSO were prepared. The solution was then serially diluted for a total of 10 serial dilutions. Dilutions were plated into the 96-well tissue culture plate. The freshly collected heparinized blood was pipetted into each well containing drug. Following a one hr incubation, Ca ionophore (0.6mM) was added to each well, stimulating the release of arachodonic acid by phospholipase A2. Diclofenac was added to each well to stop the reaction. The plates were centrifuged, to separate the cells from the plasma. The plasma was collected into an identically labeled 96-well plate, for radioimmunoassay (RIA) analysis for thromboxane B2 levels. Plotting the thromboxane counts versus the concentrations produced a sigmoidal curve. Inhibition Concentration50 (IC50) the drug dose which causes a 50% inhibition of the enzyme was then calculated.
Figure 1: COX-1 Sigmoidal curves for aspirin with IC50 determination
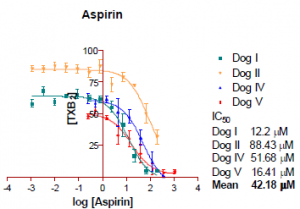
The COX-1 assay obtained reasonable IC50 values for each of the drugs sampled. Of the COX-2 assays, only repeated twice, little can reasonably be concluded without further repetitions. If initial data were repeatable it would appear that of three drugs reported, ketoprofen is the most COX-2 selective drug. To inhibit 50% of the COX-1 present it would require 42.25 times more drug than it takes to inhibit COX-2 by fifty percent.
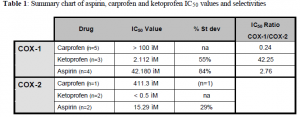
The drug with the highest COX-1/ COX-2 ratio is the ideal drug for our purposes; inhibiting COX-1 with large doses and COX-2 with low doses. By inhibiting COX-2 pain and inflammation are lessened, but by not inhibiting COX-1 the bloods clotting function can continue as normal. The results show a very curious selectivity for aspirin, a COX-2 selective ratio. Although it is not an extremely large ratio we know that in previous, but only human, studies aspirin has always been a very COX-1 selective drug. The discrepancy between species is very interesting and should be illuminated as the sequence and structure of the enzymes are elucidated and compared with the known human sequences and structures.
Ketoprofen is also aberrant, with low doses being required to inhibit COX-2. With only one repetition, however, we cannot conclusively announce ketoprofen as the new wonder drug. Once the fine-tuning of the COX-2 assay is completed there may be other drugs with higher selectivity’s for COX-2. Many repetitions remain before any findings of this study will be considered conclusive. The discovery of these unique behaviors of specific drugs in dogs is very promising, since identifying a strongly COX-2 selective drug is the ultimate goal of our research.
References
- Smith, W. l., L. J. Marnett. 1991 Review: prostaglandin endoperoxide synthase: structure and catalysis. Biochem. Et Biophys. Acta. 1083, 1-17.
- Warner, T.D.; Giullano, F.; Vojnovic, I.; Bukasa, A.; Mitchell, J.; Vane, J. Proc. Natl. Acad. Sci. 1999. 96, 7563-7568.
