Clint G. Guymon and Dr. Calvin H. Bartholomew, Chemical Engineering
The Fischer-Tropsch Synthesis (FTS) sustained Germany’s fuel needs during World War II as well as South Africa’s since the 1960’s.1 This catalytic technology is enthralling and left to the interested reader to reap in the deluge of information present in the literature. The principle reaction in FTS is a simple polymerization: nCO + 2nH2 [-CH2-]n + nH2O, over a metal such as iron or cobalt. This paper focuses on the problem of hydrothermal stability of silicasupported cobalt FTS catalyst.
It is well known that at high temperatures steam causes volatilization of silica; however, because of its formability, high surface area, and resistance to attrition it is used as a metal carrier for the said process at relatively low temperatures, 220-280 oC. Cab-O-Sil is fumed2 silica, whereas Davisil is sol-gel3 silica. These two materials are used extensively in research and industry.
We observed that the cobalt Cab-O-Sil grade M-5 catalyst irreversibly deactivated at high conversion (high partial pressures of product water) induced the conclusion that Cab-O-Sil grade M-5 is an unacceptable carrier due to its propensity to structurally transform;4 thus it was sought to show convincingly Cab-O-Sil grade M-5’s inferiority to Davisil. However, upon testing the cobalt Davisil grade 654 catalyst the same phenomenon was observed. To show that this deactivation of the catalyst was due to the collapse of the silica framework and not due to other factors such as oxidation of the metal, BET surface area measurements were taken before and after steam treatments of the two aforementioned silica carriers. After twenty-four hours of exposure to steam in a one to one molar mixture of helium and water at 220 oC and 10 atmospheres pressure the Cab-O-Sil grade M-5 lost 42% of its surface area while Davisil grade 654 lost 65% of its surface area. Thus under these conditions Cab-O-Sil grade M-5 is a more hydrothermally stable carrier; however, both lost unacceptable amounts of surface area showing that loss of activity of the cobalt catalysts can in part be earmarked to the structural changes that silica undergoes upon subjection to steam at the above conditions.
Therefore to employ the economic benefits of high conversion cobalt FTS catalysts, a new or modified carrier must be developed that has hydrothermal stability, coupled with other required characteristics, sufficiently high to justify large-scale production and implementation of said catalysts.
In continuation of the above directing events, new ideas were assimilated and developed to abate silica’s reactivity with steam by transforming the surface through the following ways: blanketing the silica with titania, hafnia, or zirconia; and transforming the morphology of the silica surface to the more stable cristobalite.
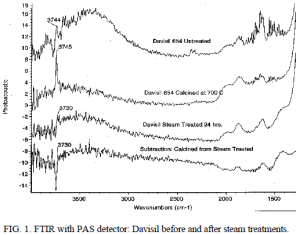
When characterizing the silica after steam treatments, a photoacoustic attachment to a Fourier Transform Infrared Spectrometer was employed to collect spectra from 4000 to 400 cm-1. We observed similar spectra as documented by other researchers including R.S. McDonald5 and Fisher et al.6 Also as documented by Fisher et al. we observed a 14 wavenumber shift of the pronounced idiosyncratic isolated hydroxyl group peak from 3745 to 3730 cm-1; Fisher et al. report a 15 point shift of the same peak from 3749 to 3734 cm-1.6 Fisher et al. state that this shift is characteristic of cristobalite formation. The above 14 cm-1 shift was observed with Davisil grade 654 after steam treatment as seen in figure 1 but not observed to occur with Cab-O-Sil grade M-5. Consequently, we plan to investigate the hydrothermal stability and physical properties of this modified Davisil 654 support.
Further study of cobalt modified silica FTS catalysts will be the focus of a master’s thesis project by the author.7
References
- Farrauto, R.J.; Bartholomew, C.H. Fundamentals of Industrial Catalytic Processes; Chapman & Hall: New York, 1997; p.342.
- See Liu, C.C.; Maciel, G.E.; J. Am. Chem. Soc. 1996, 118, 5103-5119.
- See Stober, W.; Fink, A.; Journal of Colloid and Interface Science 1968, 26, 62-69.
- Huber,G.; Bartholomew, C.H.; unpublished results, 1999, BYU.
- McDonald, R. S.; J. Am. Chem. Soc. 1957, 79, 850.
- Fisher, H. E. et al.; Inorg. Chem. 1991, 30, 4403-4408.
- The author gratefully acknowledges the aid and work of George W. Huber.