D. Jacob Butterfield, Dr. Brian Iverson, Mechanical Engineering
Introduction
This paper reports observations from condensation heat transfer tests performed on superhydrophobic (water-repelling) surfaces. Condensation is used in heat transfer applications ranging from industrial power generation to desalination. Superhydrophobic surfaces have been discovered to increase the effectiveness of this heat transfer process by increasing water droplet mobility. This project was concerned with quantifying the ability of superhydrophobic surfaces to increase the rate of heat transfer, thus making the processes condensation is used in potentially much more efficient.
Methodology
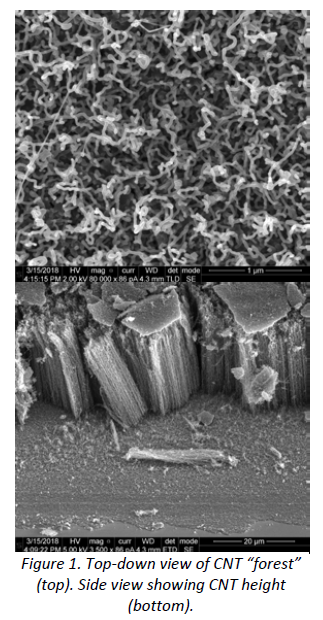
In my research project, I fabricated superhydrophobic surfaces by promoting carbon nanotube (CNT) growth on silicon and stainless steel surfaces. This was done by introducing ethylene gas on the surfaces at elevated temperatures, which produces CNT growth and creates nano-scale roughness. Vacuum pyrolysis was then performed to remove hydroxyl groups from the surfaces. The surface roughness provides fewer points for a water droplet to contact the surface and introduces gas pockets underneath the droplet which reduces friction and adhesion to the surface. I utilized Scanning-Electron Microscopy to measure CNT diameter and height (see Figure 1). The modified surface geometry, combined with the inherent hydrophobicity of carbon, renders the surfaces superhydrophobic.
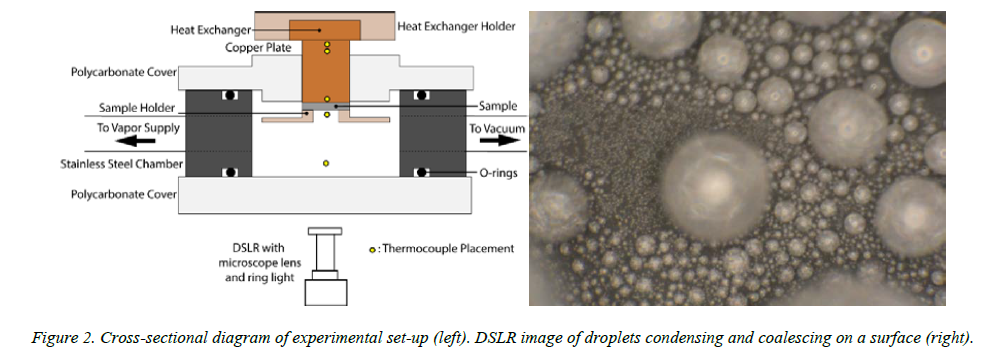
The superhydrophobic surfaces were then mounted in a vacuum chamber, shown in Figure 2 below. One important aspect of this project involved modifying the original chamber to reduce leaking and accommodate surfaces of an appropriate size. Non-condensable gases leaking into the chamber reduces heat transfer effectiveness by altering the saturation temperature at which water condenses on the surface. Essentially, an insulating layer of gas prevents the droplets from transferring heat to the cooler surface. The chamber transferred pure water vapor to the surface, which was itself cooled by chilled water, thus promoting condensation heat transfer on the surface. Thermocouples were placed throughout the chamber in order to calculate the rate of heat transfer and ensure saturated conditions.
Figure 1. Top-down view of CNT “forest” (top). Side view showing CNT height (bottom).
Results
CNT superhydrophobic surfaces were compared to hydrophilic silicon surfaces (without CNTs) in order to discover the impact that the superhydrophobic surfaces had on condensation heat transfer. Several modifications to the experimental apparatus were made in order to reduce the leak uprate, including epoxying the copper plate in place, rather than using O-ring seals. This reduced the leak uprate by an order of magnitude, thus providing much more accurate results of actual condensation heat transfer.
It was determined that contact heat resistance between the surfaces and the chiller block was one of the largest resistances to heat transfer in the setup. Despite best efforts to clamp the surfaces consistently with the sample holder (Figure 2, left image) and add a thermal grease at the interface to reduce this resistance, the difference in temperature that was used to measure the heat transfer had a higher uncertainty than the differences in heat transfer at the surface-plate interface. Visually, it appeared that more rapid condensation was promoted by coalescing droplets jumping from the CNT surfaces (Figure 2, right image). Gathering of quantitative data to reflect this difference is ongoing. Further research in this area includes quantification and reduction of the contact resistance in order to gain confidence in the absolute value of the measurements in addition to the trends. A computer vision code is also being written that tracks droplets as they form, grow, coalesce, and leave a surface, thus providing an additional method of measuring the heat transfer.
Figure 2. Cross-sectional diagram of experimental set-up (left). DSLR image of droplets condensing and coalescing on a surface (right).
Outcomes
My methods and initial results were presented at the Utah Conference for Undergraduate Research. Further, the superhydrophobic surface preparation was published in the journal Applied Physics Letters in May 2018 under the title, “Superhydrophobic, carbon-infiltrated carbon nanotubes on Si and 316L stainless steel with tunable geometry.”
Stevens, K. A., Esplin, C. D., Davis, T. M., Butterfield, D. J., Ng, P. S., Bowden, A. E., Jensen, B. D., and Iverson, B. D., 2018, “Superhydrophobic, carbon-infiltrated carbon nanotubes on Si and 316L stainless steel with tunable geometry,” Applied Physics Letters, Vol. 112, pp. 211602.