George Major and Faculty Mentor: Dr. Matthew Linford, Department of Chemistry and Biochemistry
Thin Layer Chromatography (TLC) is widely used for performing chemical separations and has been a staple in separation techniques. However, the functionality and effectivity of TLC plates has not changed over the past several decades. Standard TLC plates have a slow developing time compared to other chemical separation methods, rendering the technique a potential bottleneck in industrial processes. TLC plates are ripe for innovation to provide faster separations and better resolution. The Linford group, over the past few years, has been developing a novel method for creating a suitable TLC plate using a micropatterned silica wafer using SU-8, a negative photoresist, with photolithography for creating a suitable mold to deposit prepared silica (fig. 1). A major step in creating the plate is developing a method to deposit silica 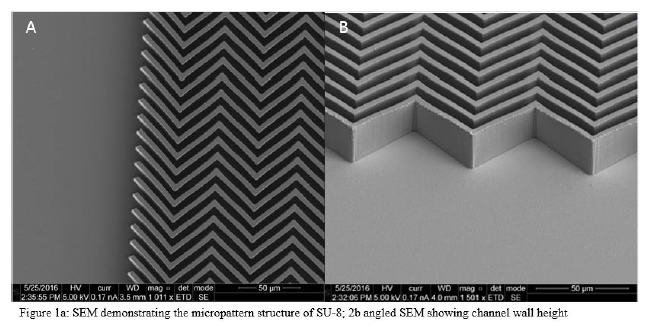 into the micropattern channels and then remove t
into the micropattern channels and then remove t
he photoresist to leave channels of silica, suitable for TLC. The project has successfully deposited mesoporous silica into microchannels and successfully removed the SU-8 and functionalized the silica remaining on the wafer. The measured height of the final silica walls of the channel are ca. 25μm tall, with channel width being 6μm. Energy dispersive spectroscopy (EDS) data shows complete removal of carbon, indicative of complete removal of SU-8. SEM images confirm appropriate channel filling and structural pattern retention.
Methods
Silica nanoparticles (ca. 40nm diameter) were prepared into a slurry using 20wt/% using millipore water, with 2wt/% polyvinyl alcohol (PVA) as a binder. PVA was chosen as a binder over gypsum because of the leeching properties of HF, which is to be used later in the industrial process to functionalize the silica after removal of the SU-8. There
fore, an organic alcohol binder is used rather than calcium sulfate. Immediately before application, methanol (5vol/%) is added to the slurry to promote surface wetting. The micropatterned, SU-8 silica wafer is plasma treated for 40 seconds to help functionalize the surface and expose hydroxyl groups, as well as to remove the thin 2-3nm thick layer of Omnicoat used for adhesion promotion during photolithography. This reveals bare silica in the channels which is necessary for a later process to create siloxane bonds between the substrate and the slurry. Using a micropipette, 1-1.5mL slurry
is deposited onto a silica, micropatterned plate 2.5x10cm. A rubber squeegee is wetted with methanol and used to pull the slurry down over the channels and remove any excess slurry from the top of the channels. The squeegee is used only twice to pass over the plate, being washed with methanol between each pass. After deposition, the plates
dry overnight in atmosphere. Figure 2 shows the SEM results of channels filled with the silica nanoparticles. Note the visible tops of the SU-8 channels. Improper passing with the squeegee would allow excess silica to remain on top, and not allow channels to be created (figure 2c). The plate is then heated to 900°C for 2 hours under inert atmosphere of Argon. This is to slow the removal of organics and allow the formation of siloxane bonds to form before the removal of the SU-8 pattern and the PVA binder. Our experiments have shown that heating under atmosphere causes collapse of the silica structure. This may be because the temperature required to remove SU-8 is ca. 300°C, where the temperature required to begin siloxane binding is around 880°C.

Discussion
Through the process of developing this method to deposit silica into an organic photoresist pattern, many variables and considerations were required to meet the needs of the overarching project. The details of the project and the many iterations it went through before this final method is evident even compared to the initial proposal where silica nanoparticles and PVA were not even considered as an option for the slurry. The choice of a squeegee to remove and deposit the silica is prone to human error, but is industrially feasible and therefore remained a part of the procedure. The initial processes that were considered were much more step intensive, and although the project saw a lot of preliminary success, creating a procedure that only required a few simple steps with industry in mind proved to be a much larger challenge. In addition to these considerations, as mentioned above, chemical restrictions were in place due to the subsequent steps to functionalize the silica plates using HF, which removes ions from substrates. Initial testing using the gypsum binder proved successful until this step, which is not directly a part of this project, which removed all the silica structure from the wafer, rendering the project useless. Because of this constraint, a new binder was required and a new slurry ratio was created and optimized. There is no published literature the Linford group could find for depositing silica into a photopatterned microstructure as described here. This is a completely novel design and has shown to be successful for the overall purpose of the creation of a novel TLC plate, as well as for the purpose of expanding the use of photolithography with practical, microstructure design.
