Bair, Stephanie
The Effect of Chronic Ethanol on VTA GABA Neurons
Faculty Mentor: Scott Steffensen, Psychology
Introduction
The current accepted model for addiction in the brain is the dysregulation of dopamine (DA) in the mesolimbic pathway, which begins in the ventral tegmental area (VTA) and projects to the nucleus accumbens (NAc). DA neurons originate in the VTA and project to the NAc where they release DA. Dopamine neurons are typically regulated locally by inhibitory gamma-aminobutyric acid (GABA) neurons in the VTA. Because GABA neurons in the VTA regulate DA neurons, we conclude that changes in VTA GABA neurons will affect DA release in the NAc underlying addictive behaviors, especially reward seeking.
We have examined the synaptic plasticity of GABAergic synapses on VTA GABA neurons to better understand their role in the electrical activity of GABA neurons. Plasticity is often thought to be primarily due to glutamate (GLU) transmission; however, plasticity refers to any long-term changes in the cell, and can be mediated by GABAergic synapses as well (Nugent & Kauer 2008). Previous research has shown that VTA GABA neurons become hyperexcitable during withdrawal from ethanol (Gallegos et al., 1999), and our experiment has investigated whether the synaptic plasticity seen with ethanol is due to GABAergic synapses.
Methodology
We will be using patch-clamp electrophysiology techniques in brain slice preparations to measure synaptic inputs into VTA GABA cells. In order to identify GABA neurons, we used GAD GFP knock-in mice, with a genetic modification causing the GABA neurons in a slice to fluoresce under UV light. We then record the inhibitory (IPSC) and excitatory (EPSC) currents that contribute to the depolarization or hyperpolarization of the cell. We will use the GLU receptor antagonist kynurenic acid to block EPSCs so that we are selectively isolating IPSCs. Using just kynurenic acid will allow us to measure spontaneous IPSCs (sIPSC), but we will also use lidocaine, a voltage-gated sodium channel blocker, to block event related potentials so we can record mini IPSCs (mIPSC).
For the chronic condition, we placed mice in chronic intermittent ethanol (CIE) vapor chambers designed to expose mice to ethanol vapor for a period of 16 hours per day for 3 weeks. Then we recorded IPSCs 24 hours after the last exposure to ethanol. Control mice were placed in the same chambers and followed the same procedure, but were exposed to air in place of ethanol. For the acute ethanol condition, we performed an intraperitoneal (IP) injection of 4.0g/kg ethanol 24 hours before performing experiments.
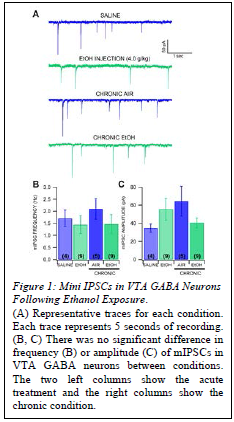
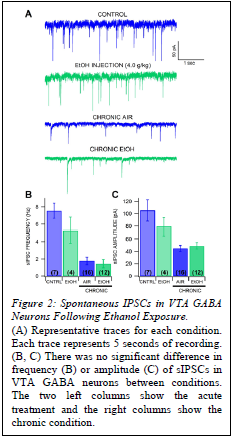
Results
mIPSCs were measured as a sign of GABA plasticity (Fig. 1). There was no significant difference in mIPSC frequency in VTA GABA neurons between animals that received a single injection of ethanol (4.0 g/kg) and saline-injected controls or between CIE vapor-exposed mice and air-exposed controls. There was no significant difference in amplitude of mIPSCs between mice that received a single injection of ethanol and saline-injected controls, and no significant difference between CIE vapor-exposed mice and air-exposed mice.
sIPSCs were measured as a sign of GABA plasticity (Fig.2).. There was no significant difference in sIPSC frequency in VTA GABA neurons between mice that received a single injection of ethanol (4.0 g/kg) and controls, or between air-exposed and CIE vapor-exposed mice in the chronic condition. There was also no significant difference in amplitude of sIPSCs between mice that received a single injection of ethanol and controls, or between air-exposed and CIE vapor-exposed mice.
Discussion
We found no significant difference in the inhibitory synaptic events between groups in either the acute or chronic condition. While these results were not what we hypothesized, they do not entirely rule out the role of GABA plasticity in the chronic condition, and they allow for the possibility of glutamatergic plasticity causing the shift in VTA GABA neurons to become excitable that was stated previously. Additionally, the possibility exists that the function of these inhibitory synaptic events is changing rather than a change in frequency or amplitude. In our future studies, hopefully we can investigate the role of GLU plasticity in VTA GABA neurons after alcohol exposure to better explain the changes that are occurring, especially in the acute condition.
Conclusion
We aimed to understand how GABA plasticity contributes to the changes we have seen previously in VTA GABA neurons in response to ethanol that lead to the dysregulation of DA neurons leading to dependence and addiction. Our results were not statistically significant or entirely conclusive to either support or reject our hypothesis. However, these results will lead us to further investigations about the physiological changes that occur to produce the dependent state in the brain.
Sources
Nugent, F. S., & Kauer, J. A. (2008). LTP of GABAergic synapses in the ventral tegmental area and beyond. The Journal of physiology, 586(6), 1487-1493.
Gallegos, R. A., J. R. Criado, et al. (1999). “Adaptive responses of GABAergic neurons in the ventral tegmental area to chronic ethanol.” J. Pharmacol. Exp. Ther. 291: 1045-1053.