Broadbent, James
Elucidation of Mechanisms Whereby Embryonic Chicken Limbs Develop
Faculty Mentor: Jeffrey Barrow, Department of Physiology and Developmental Biology
Introduction
As established by John Saunders 60 years ago, the AER is required for limb growth and patterning. However, the mechanisms whereby the AER regulates these processes remain poorly understood. My professor and I propose that one of the primary roles of the AER is to regulate directional growth of the adjacent limb mesenchyme. Over time, the AER extends along the anteroposterior (AP) and thins along the dorsoventral (DV) axes. These results highlight a morphogenetic paradigm: the dimensions of recruitment signaling centers ultimately shape organs. This model also provides mechanistic insight for evolutionary change. We also show that sonic hedgehog (Shh) protein, excreted by the zone of polarizing activity (ZPA), directly regulates AER viability. The ZPA regulates AER viability through Shh signaling in the posterior AER. Surgical removal of the posterior AER and implantation of a Shh-soaked bead in the anterior mesenchyme maintains anterior AER.
Methodology
• Surgical micro needles for making incisions in chick embryos.
• Forceps to aid in surgical processes.
• Agarose micro beads to soak up either Shh or PBS. Used in bead insertion experiments.
• Shh (sonic hedgehog) protein stock.
• Nile Blue stain for AER identification.
• Phosphate-buffered saline solution (PBS).
• Fresh stock of fertilized chicken eggs each week.
• Incubator
Results & Discussion
Figure 1. Recovery of the AER after partial surgical removal. (A-F) The AER of Stage 21 limbs was stained with Nile Blue and portions thereof were removed (i.e., anterior half (A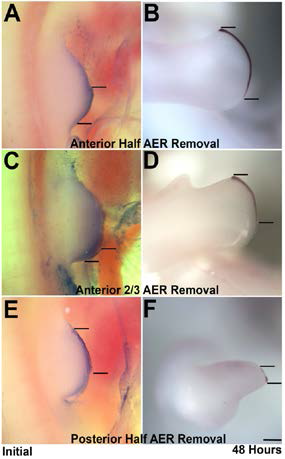 ), anterior 2/3 (C), posterior 1/2 (E). The limb buds were photographed and the length of the AER measured. After a 24-48 hour incubation the embryos were fixed and subjected to Fgf8 in situ hybridization (B,D,E). The stained AER was measured. As can be seen remnants of the posterior AER double in length over a 48-hour period (A-D) whereas anterior remnants shrink over time. Scale bar = 300 mm.
), anterior 2/3 (C), posterior 1/2 (E). The limb buds were photographed and the length of the AER measured. After a 24-48 hour incubation the embryos were fixed and subjected to Fgf8 in situ hybridization (B,D,E). The stained AER was measured. As can be seen remnants of the posterior AER double in length over a 48-hour period (A-D) whereas anterior remnants shrink over time. Scale bar = 300 mm.
Figure 2. Shh is responsible for AER viability. (A,B) Posterior AER is surgically removed. PBS-soaked beads are
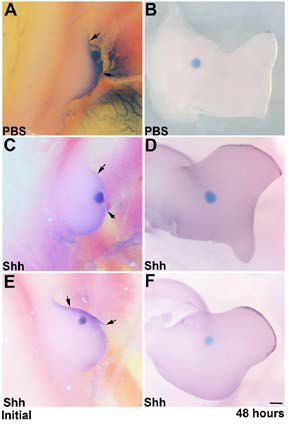 implanted in the mesenchyme of the anterior limb bud. Anterior AER disappears after a 48-hour incubation. (C-F) Posterior AER is removed and Shh-soaked beads are implanted in the mesenchyme of the anterior limb bud. After 48 hours, anterior AER is maintained.
implanted in the mesenchyme of the anterior limb bud. Anterior AER disappears after a 48-hour incubation. (C-F) Posterior AER is removed and Shh-soaked beads are implanted in the mesenchyme of the anterior limb bud. After 48 hours, anterior AER is maintained.
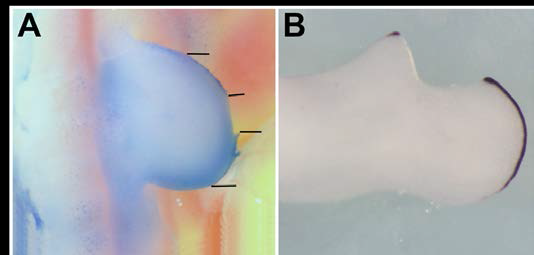
Figure 3. Central AER removal results in anterior AER deterioration. (A,B) A central portion of the AER is surgically removed. Over 48 hours, anterior AER shrinks significantly and posterior AER fills the central area.
Conclusion
The AER is necessary and sufficient for directed growth of the mesenchyme (bone precursor cells). AER dimensions dictate the shape of the limb mesenchyme that follows. AER shape changes dramatically over time. Initially, it is short along the AP axis and thick long the DV axis consistent with mesenchyme recruiting that would give rise to the stylopod. Later, it extends along the AP and thins along the DV axes to recruit mesenchyme, corresponding to the shape of the distal zeugopod and autopod elements. Removal of the anterior half of the AER results in regrowth of the excised AER. After removal of the posterior half of the AER, anterior AER disappears. (Fig. 6) Shh is responsible for AER viability. (Fig. 7) Removal of a central portion of the AER results in anterior AER disappearance, and posterior AER regrowth. (Fig. 8) The positive response of surgically-altered AER to Shh placement demonstrates the interdependence of Shh production, AER viability, and ultimately three-dimensional limb shape.