Peck, Connor
The Chemotherapeutic Potential of Resveratrol-Based Treatments
Faculty Mentor: Kim O’Neill, Department of Microbiology and Molecular Biology
Introduction
Cancer is one of the most prevalent and deadly diseases in the world. In 2012 alone, there were
approximately 14 million new cases of cancer and over 8 million cancer-related deaths
worldwide. Despite extensive research in past decades, the burden of cancer continues to
increase.
One of the most common treatments for cancer is chemotherapy, the use of various chemical
substances that exhibit anti-cancer activity. Unfortunately, chemotherapy can be quite toxic and
often produces inadequate results. There remains a great need for improved chemotherapeutic
treatment strategies in order to improve outcomes for cancer patients.
The purpose of my project was to investigate the chemotherapeutic potential of an anti-oxidant
known as resveratrol. Resveratrol is a compound naturally found in many fruits such as grapes
and blueberries. Resveratrol is safe for normal cells in our bodies, but has actually been shown to
be toxic to cancer cells. Nonetheless, its potential for therapeutic use continues to be highly
debated. In our study, we wanted to see if the effects of chemotherapy on cancer cells could be
enhanced by co-treatment with low levels of resveratrol.
We also wanted to test the chemotherapeutic potential of an analog of resveratrol called
pterostilbene. Previous results in our lab show that pterostilbene might also possess anti-cancer
activity, but relatively little research has been done on the compound up to this point.
Methodology
First, we grew a cancerous lymphoma cell line. We then treated these cells with 5-Fluorouracil
(5-FU), a commonly used chemotherapy drug. We also treated a portion of the cells with either
resveratrol or pterostilbene. Other cells were treated with a combination of both compounds.
This allowed us to compare normal chemotherapy treatment to treatments with one or both of
these compounds added.
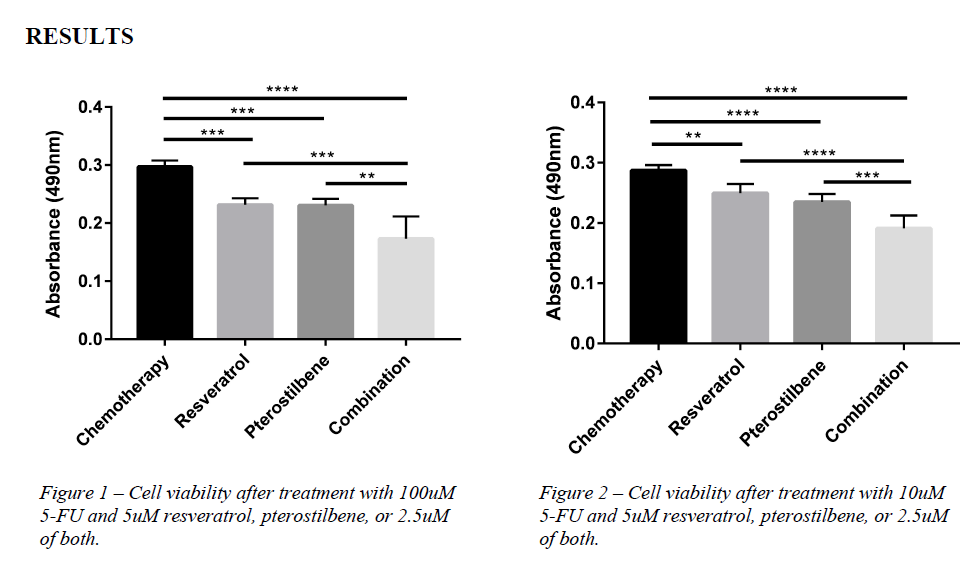
We then used an assay called an MTS assay to measure the viability of the cells treated—that is,
how many cells were killed by the chemotherapy. In this assay, viable cells are able to produce a
fluorescent product that can be measured. The results of this assay are shown below.
The second step of this project was to figure out what mechanisms were at work to produce the
results shown in Figures 1 and 2. Unfortunately, we have been unsuccessful in doing so. We
have tried several techniques but have suffered many setbacks in the lab. We are currently
working to answer this question.
Our results, shown in Figure 1 and Figure 2, indicate that co-treating cells with either resveratrol
or pterostilbene significantly increased the toxicity of 5-FU at both 100uM and 10uM. Viability
of cells after treatment with the compounds was decreased significantly in comparison to
viability after treatment with just chemotherapy. Combinations of pterostilbene and resveratrol
further enhanced 5-FU toxicity.
Discussion
These results support the potential use of resveratrol and pterostilbene in chemotherapy, as both
compounds clearly enhanced 5-FU toxicity to B-cell lymphoma even at low, physiologically
possible concentrations. Interestingly, combinations of the compounds were even more effective.
This suggests that the two compounds may act to activate cell death in different ways—
something that remains to be inspected. While these results are still quite preliminary, they
clearly demonstrate the chemotherapeutic potential of both compounds and that warrants further
studies.
Conclusion
Our results show that both resveratrol and pterostilbene possess anti-cancer activity and indicate
their great potential for use in chemotherapy. There is, however, much work that remains to be
done in order to better characterize the effects of these compounds. There have been some
unfortunate drawbacks that have slowed this project down, but we are proud of the work that has
been done.