Brandon Gassaway and Dr. Emily Bates, Chemistry and Biochemistry
Anderson-Tawil Syndrome (ATS) is characterized by morphological defects including clinodactyly (abnormal curvature of digits), syndactyly (fused digits), micrognathia (small jaw), cleft palate, and hypertelorism (wide-set eyes) as well as periodic paralysis and heart arrhythmia. Mutations in inwardly rectifying potassium (Irk) channels are found in patients with ATS. Expression of mutant Irk channels causes similar physiological and morphological defects in mice.
Irk channels are transmembrane proteins consisting of four subunits that coordinate together to form an ion conduction pathway across the cell membrane. The genetic mutation in Irk channels that causes ATS creates a dominant negative product, or a protein that lacks or contains a disrupted functional domain, but is still able to form the tetramer. The presence of one dominant negative subunit in the tetramer may be sufficient to alter proper ion conduction, and causes the paralysis and arrhythmia characteristic of ATS.
Periodic paralysis and heart arrhythmia are logical consequences of mutations in Irk channels that regulate ion flow across cell membranes and neuronal membrane excitability. The morphological defects observed, however, were surprising because they suggest a previously unknown link between membrane excitability and development. Preliminary data using a dominant negative allele of Irk 2 suggests that Irk 2 function is necessary for embryogenesis and adult development in Drosophila. This project characterized these roles by analysis of Irk 2 knockout alleles and genetic interaction studies.
Decapentaplegic (Dpp) is a member of the Transforming Growth Factor ß superfamily. The Dpp pathway is involved in dorsal/ventral (front/back) signaling during development. The protein Dpp is a morphogen that binds to a complex made up of type I and type II serine threonine kinase receptors thickveins (Tkv) and Punt. Disruption of Irk 2 function with a dominant negative allele produces similar phenotypes to those observed in Drosophila with defects in the Dpp pathway. Thus, the secondary aim of this project was to characterize a putative interaction between the Irk 2 channel and the Dpp pathway.
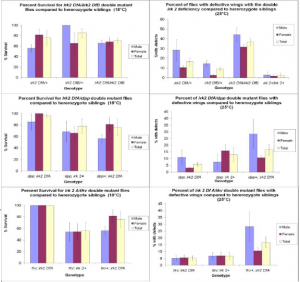
To determine the requirement for Irk 2 in development, two Irk 2 deletion alleles were generated.
These two deletion alleles have different deletion boundaries, allowing the generation of flies that lack Irk 2 function entirely without removing function of more than 5 other surrounding genes. Irk 2 knockout animals were compared to animals that are heterozygous for deletion alleles and to wild type animals, and characterized by percent survival and morphological defects in the wings.
To determine if Irk 2 plays a role in the Dpp pathway, Irk 2; dpp and Irk 2; tkv7 double mutants were generated and characterized. Double mutants were compared to flies with one Irk 2 deficient allele and wildtype animals. We looked for either suppression or enhancement of Irk2 phenotypes by Dpp components. The phenotypes quantified from this cross are the same as the Irk 2 knockout studies; percent survival and morphological defects in the wings.
In the analysis of the knockout animals, we determined that Irk 2 was necessary for development. Knockout flies have a significantly higher percentage of defective wing phenotypes than their heterozygote siblings (irk2 Df/+) (fig. 1). In the control experiment, knockout flies had almost a 10 fold higher occurrence of wing defects when compared to wild type flies. The knockout animals also had a lower rate of survivability than their heterozygote siblings. This data suggests that these defects are caused by the Irk 2 gene.
A putative interaction between Irk 2 channels and the Dpp pathway was also supported by the data, which showed suppression of the defective phenotype in both the dpp; irk2 Df and tkv; irk2 Df double mutants. These double mutants had higher rates of survivability and lower percentages of defective wing phenotypes than their Irk 2 deficient only siblings. If the data had indicated a worsening effect, it would not have necessarily indicated an interaction, as the two detrimental effects could have simply been additive. If the data had shown no effect, then either there was no interaction, or the two alleles counteracted each other. However, the only way for the phenotype to be suppressed would be if the two genes interacted. Thus, the suppression of the defective phenotype demonstrates an interaction between Irk 2 channels and the Dpp pathway.
In the future, electrophysiological and epistatic techniques will be used to characterize the interaction between Irk 2 and Dpp, and eventually determine the role of Irk 2 channels in the Dpp pathway.
Fig. 1 a. Irk 2 knockout flies show a lower survival rate at 18°C and higher percentage with wing defects at 25°C than heterozygous siblings. b. Suppression of the defective phenotype from irk 2A/dpp and irk 2A/tkv.