Tyler Ekins and Scott Steffensen, Psychology
Introduction
Methamphetamine (METH) abuse is a problem that continues to cause much harm across the United States. The cost of METH use in 2005 alone was estimated to be 23.4 billion dollars (Nicosia et al., 2009). Other societal costs of METH abuse include suicide attempts, premature death, increased health care expenses, loss of workplace productivity, incarceration, child maltreatment, foster care, violent crime, and theft (Nicosia et al., 2009). Understanding the mechanism by which METH acts on the brain is the first step in being able to synthesize a pharmacotherapeutic method to treat METH addiction.
Part of the reason that METH is so rewarding, and therefore abused, is that it dramatically increases dopamine (DA) levels in the nucleus accumbens (NAc), a subarea of the striatum that has been implicated in natural and drug reward. METH acts on two separate DA transporter systems: DA transporters (DAT), located on outer membranes of presynaptic DA neurons, and type-2 vesicular monoamine transporters (VMAT-2), located on synaptic vesicles inside presynaptic DA neurons. METH binds to the DAT and reverses its function, pumping DA into the synaptic cleft (Ritz et al). METH is also thought to bind to VMAT-2 as a competitive inhibitor, blocking DA from being repackaged into vesicles (Brown et al). Increased levels of unpackaged DA in the cytosol can spill through the DAT into the synaptic cleft, further increasing DA levels and signaling.
DA is highly reactive and is subject to numerous regulatory pathways. It can be metabolized, stored in vesicles or converted to neuromelanin. Cytoplasmic or extracellular DA is converted to DOPAL by monoamine oxidase. Hydrogen peroxide and hydroxide ion are produced in the process, creating reactive oxygen species (ROS). Additionally, free DA can auto-oxidize and create more ROS. ROS are reactive chemical compounds and are involved in many normal cellular functions such as cell signaling and oxygen metabolism. However, when ROS levels are inordinately high they produce oxidative stress, which can damage cell machinery.
TEMPOL (4-hydroxy-2,2,6,6-tetramethylpiperidine-1-oxyl) is a superoxide dismutase (SOD) mimetic, which is a specific ROS scavenger; it seeks out and binds to superoxide, rendering it inert. N-acetylcysteine (N-Cys) stimulates production of glutathione (GSH), an important intracellular antioxidant that is unable to cross the blood-brain barrier (BBB). N-Cys, however, is able to cross, and once inside neurons, is enzymatically converted into GSH. TEMPOL and N-Cys have been known to attenuate cocaineinduced DA release and cocaine-taking behavior (Jang et al., 2014; Numa et al., 2008). Because METH is a psychostimulant that acts pharmacologically similar to cocaine, we proposed that TEMPOL and N-Cys should similarly decrease METH-induced DA release in the brain.
Methodology
Immunohistochemistry
We obtained coronal brain slices containing the nucleus accumbens stained with 8-Hydroxyguanosine (8- OHG) fluorescent antibodies from groups of male Wistar rats injected with either saline or METH. 8- OHG antibodies target a modified base that occurs in DNA/RNA as a result of oxidative stress. The brain slices were analyzed using confocal microscopy.
Fast Scan Cyclic Voltammetry
For in vivo recording, carbon fiber microelectrodes were filled with 3M KCl and implanted into the nucleus accumbens core. Dopamine release was evoked by a 4.0 msec, ten-pulse electrical stimulation from a bipolar, coated stainless steel electrode stereotaxically implanted into the medial forebrain bundle. Collections were taken periodically every 2 minutes throughout the course of each experiment.
TEMPOL and N-Cys Administration
Following a stable control (i.e. baseline DA level), TEMPOL or N-Cys was administered via intraperitoneal (IP) injection. Collections were taken for 10 minutes, and then METH (0.1 mg/kg, IV) was administered through a jugular vein catheter and collections were taken for 1 hour.
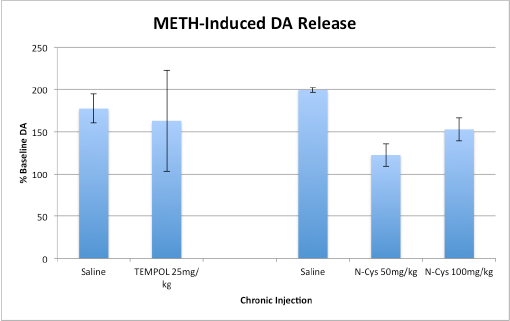
Groups of rats were pretreated with either saline, TEMPOL or N-Cys. The pretreatment consisted of 1 IP injection daily for 5 days prior to the in vivo recording. The groups were as follows: TEMPOL 25 mg/kg (n=4) compared to Saline (n=5) and N-Cys 50 mg/kg (n=2) and N-Cys 100 mg/kg (n=4) compared to saline: (n=5).
Results
Results from immunohistochemistry and confocal analysis confirm that METH increases oxidative stress in the NAc.
Chronic TEMPOL administration had no significant effect on attenuation of METH-induced DA release in the NAc (Saline; n = 5 (177.5±17.2), TEMPOL 25 mg/kg; n = 4 (162.7±59.7)).
Chronic N-Cys administration, however, successfully reduced METH-induced DA release, and had more powerful effects at lower doses of the drug (N-Cys 50 mg/kg; n = 2 (122.3±13.3), N-Cys 100 mg/kg; n = 4 (152.7±13.4), Saline; n = 5 (199.2±3.0)).
Discussion
As previously mentioned, METH acts on VMAT and DAT to increase extracellular DA concentration and produce ROS. It is unclear why TEMPOL works to attenuate cocaine but not METH-induced DA release, but it is possible that VMAT is involved, as cocaine acts on DAT but not VMAT. Additionally, TEMPOL is an SOD mimetic, meaning that it is a more specific kind of ROS scavenger. METH produces a massive increase in extracellular DA, and therefore it is likely that METH produces many different types of ROS, not simply superoxide. TEMPOL could be too specific to significantly reduce ROS levels.
N-Cys is a more general antioxidant; it binds to more than just superoxide, and was effective in attenuating METH-induced DA release. Doses of chronically injected N-Cys may be toxic to the animal at high levels, which would confirm the finding that N-Cys was a more effective antioxidant at lower doses. It should be noted that only 2 rats were used at the dose of 50 mg/kg; more experiments need to be done at this dose.
Conclusion
This study provides evidence that ROS are involved in the mechanism of action of METH. We have shown that chronically injected N-Cys but not TEMPOL successfully tends to attenuate METH-induced DA release in the NAc. The exact mechanism by which ROS increase DA release still remains unclear, however these data show that there is a connection, as removing ROS decreases DA release. This study is one further step in the direction of understanding how METH acts on the brain, which is a prerequisite to treat METH addiction.
References
- Brown, J. M., Riddle, E. L., Sandoval, V., Weston, R. K., Hanson, J. E., Crosby, M. J., . . . Fleckenstein, A. E. (2002). A single methamphetamine administration rapidly decreases vesicular dopamine uptake. J Pharmacol Exp Ther, 302(2), 497-501.
- Jang, E., Ryu, Y., Lee, B., Chang, S., Yeo, M., Kim, S., et al. (2014). Involvement of reactive oxygen species in cocaine-taking behaviors in rats. Addict Biol, 10.1111/adb. 12159.
- Nicosia, N., Pacula, R.L., Kilmer, B., Lundberg, R., Chiesa, J. (2009). The Economic Cost of Methamphetamine Use in the United States, Rand.
- Numa, R., Kohen, R., Poltyrev, T., & Yaka, R. (2008). Tempol diminishes cocaine-induced oxidative damage and attenuates the development and expression of behavioral sensitization. Neuroscience,155(3), 649-58.
- Ritz, M. C., Lamb, R. J., Goldberg, S. R., & Kuhar, M. J. (1987). Cocaine receptors on dopamine transporters are related to self-administration of cocaine. Science, 237(4819), 1219- 1223.