Matthew Landeen and Dr. Alonzo Cook, Chemical Engineering
Introduction
The neurobiological and biochemical processes of cellular degeneration, secondary to trauma in the peripheral nervous system is well documented; however the regenerative potential of the peripheral nervous system is still under intense scrutiny. In this study, the rate of regeneration of the rat sciatic nerve was measured after intraneural injection treatment of porcine UBM (urinary bladder matrix). It is well documented that UBM (MatriStem) (ACell Inc.) has therapeutic effects on tissue reconstruction and outgrowth. Results from this study were compared to our previous study including intraneural injections of NGF (nerve growth factor), LPC (lysophosphatidycholine), and a combination treatment including both NGF and LPC. Regeneration was measured using electrophysiology for quantitative analysis. Methods of qualitative analysis included histology and SEM imaging.
Methods
In the present study, isogenic female Wistar Albino rats were divided into 3- week and 6-week groups. All rats received the same treatment of UBM. Prior to surgery, rats were weighed, and anesthetized using Isoflurane. Once the rats were properly sedated, an intraperitoneal injection of Buprenorphine was administered, using a 27 gauge needle. Prior to transferring the rats to the operating table, the left hind leg was shaven. The rats were then transferred to the operating table, and three sets of an alternating series of Iodine and 70% isopropyl alcohol was used to disinfect the surgical site. Clear sterilized drape clothes with a circular opening were then placed over the animal’s hind leg, to maintain the integrity of the disinfected field. Finally, a surgical marker was used to mark the anatomical incision site so as to maintain a standardized procedure between surgeons. The animal’s hind leg skin was incised approximately 5mm caudal to the femur, identified by palpation. The incision was made parallel to the femur, and the facial plane between the piriformis and quatratus femoris muscles was penetrated by reverse scissor technique. Once the sciatic nerve was visible, it was raised from the cavity using a glass hook, and the nerve was crushed using forceps. The crush procedure consisted of a single 20 second crush followed by a single 10 second crush. Incisions were closed using sterilized wound clips. Rats were monitored and treated for pain according to protocol received from the veterinarian.
Seven days after the crush procedure was performed, UBM was prepared and injected directly into the sciatic nerve using the documented needles with a manipulator and a microscope at 40X magnification. The UBM concentration was 4mg/mL in a solution of 0.9% isotonic sodium chloride with fast green for visual verification. Electrophysiology analysis was performed three days a week for both 3-week and 6-week groups. After this time the rats were euthanized. Upon euthanasia, histology and SEM imaging was gathered for qualitative analysis.
Results
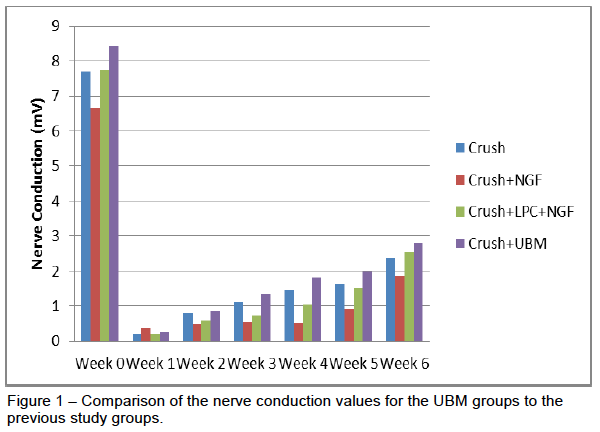
There were 5 rats in the 3-week group and 6 rats in the 6-week group. Each group started off with 9 rats but due to self mutilation 7 of the rats had to be euthanized early. The UBM treatment performed very similar to our previous study. From our electrophysiology data, the rats had a significant decline in their nerve conduction after receiving a crush injury. The conduction dropped to around 2% of the pre-surgery values and steadily rose over the course of the next six weeks to achieve about 33% of the pre-surgery values. As seen in Figure 1, the UBM achieved a higher nerve conduction average than any of the other groups previously tested. The rats were split into a 3-week and 6-week for immunohistochemistry and nerve imaging. This work is still being processed.
Discussion
The relative rate for regeneration in the peripheral nervous system in general is slow. In our 6-week group of rats we started to see a trend of increased rates of regeneration around week six for those receiving the UBM treatment compared to those in other groups. The rats in this group may show significant regeneration if additional studies measure regeneration for a longer period of time. UBM contains many proteins other than ones necessary for regeneration. Not much is known about the interaction of these proteins. If the proteins necessary for regeneration could be isolated we might see better results. The electrophysiology we performed might be interfering with regeneration as well. The electricity used to stimulate the muscle could be providing a regenerative effect for all groups making our data less significant for our desired measures.
Conclusion
We are still analyzing data from the axon diameters, number of axons and number of myelinated axons. We hope to gain additional information on the regenerative effects of UBM and a better understanding on how to effectively measure regeneration. Our research has been presented at many conferences and we are working on submitting a paper for publication. I would like to thank Alan Harker for the ORCA grant, ACell Inc. for providing their UBM product MatriStem and Dr. Cook for mentoring me through this project.